A Study in Scaffold Hopping: Discovery and Optimization of Thiazolopyridines as Potent Herbicides That Inhibit Acyl-ACP Thioesterase.
Abel, S.A.G., Alnafta, N., Asmus, E., Bollenbach-Wahl, B., Braun, R., Dittgen, J., Endler, A., Frackenpohl, J., Freigang, J., Gatzweiler, E., Heinemann, I., Helmke, H., Laber, B., Lange, G., Machettira, A., McArthur, G., Muller, T., Odaybat, M., Reingruber, A.M., Roth, S., Rosinger, C.H., Schmutzler, D., Schulte, W., Stoppel, R., Tiebes, J., Volpin, G., Barber, D.M.(2023) J Agric Food Chem 71: 18212-18226
- PubMed: 37677080 Search on PubMed
- DOI: https://doi.org/10.1021/acs.jafc.3c02490
- Primary Citation Related Structures:
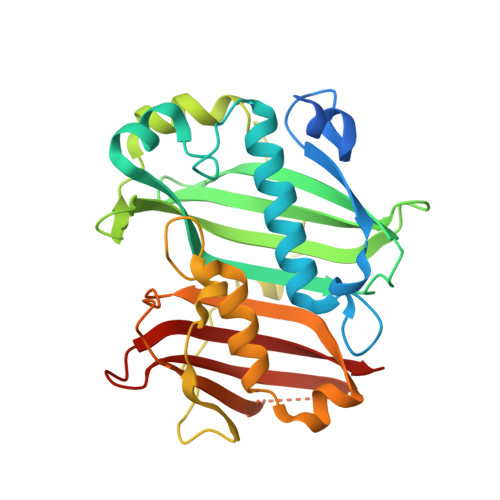
8P8K - PubMed Abstract:
In the search for new chemical entities that can control resistant weeds by addressing novel modes of action (MoAs), we were interested in further exploring a compound class that contained a 1,8-naphthyridine core. By leveraging scaffold hopping methodologies, we were able to discover the new thiazolopyridine compound class that act as potent herbicidal molecules. Further biochemical investigations allowed us to identify that the thiazolopyridines inhibit acyl-acyl carrier protein (ACP) thioesterase (FAT), with this being further confirmed via an X-ray cocrystal structure. Greenhouse trials revealed that the thiazolopyridines display excellent control of grass weed species in pre-emergence application coupled with dose response windows that enable partial selectivity in certain crops.
- Research and Development, Weed Control Research, Bayer AG, Crop Science Division, Industriepark Höchst, 65926, Frankfurt am Main, Germany.
Organizational Affiliation: