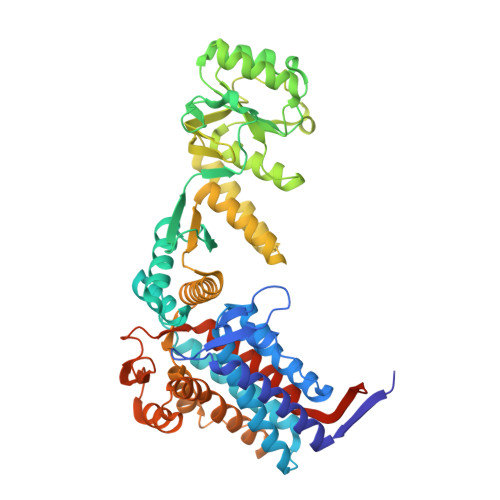
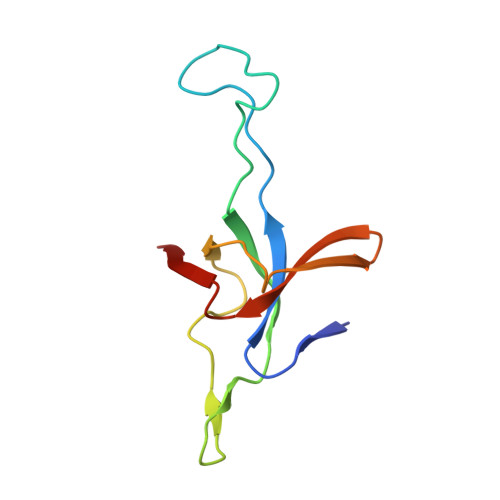
Visualizing chaperonin function in situ by cryo-electron tomography.
Wagner, J., Carvajal, A.I., Bracher, A., Beck, F., Wan, W., Bohn, S., Korner, R., Baumeister, W., Fernandez-Busnadiego, R., Hartl, F.U.(2024) Nature 633: 459-464
- PubMed: 39169181 Search on PubMedSearch on PubMed Central
- DOI: https://doi.org/10.1038/s41586-024-07843-w
- Primary Citation Related Structures:
8P4M, 8P4N, 8P4O, 8P4P, 8P4R, 8QXS, 8QXT, 8QXU, 8QXV - PubMed Abstract:
Chaperonins are large barrel-shaped complexes that mediate ATP-dependent protein folding 1-3 . The bacterial chaperonin GroEL forms juxtaposed rings that bind unfolded protein and the lid-shaped cofactor GroES at their apertures. In vitro analyses of the chaperonin reaction have shown that substrate protein folds, unimpaired by aggregation, while transiently encapsulated in the GroEL central cavity by GroES 4-6 . To determine the functional stoichiometry of GroEL, GroES and client protein in situ, here we visualized chaperonin complexes in their natural cellular environment using cryo-electron tomography. We find that, under various growth conditions, around 55-70% of GroEL binds GroES asymmetrically on one ring, with the remainder populating symmetrical complexes. Bound substrate protein is detected on the free ring of the asymmetrical complex, defining the substrate acceptor state. In situ analysis of GroEL-GroES chambers, validated by high-resolution structures obtained in vitro, showed the presence of encapsulated substrate protein in a folded state before release into the cytosol. Based on a comprehensive quantification and conformational analysis of chaperonin complexes, we propose a GroEL-GroES reaction cycle that consists of linked asymmetrical and symmetrical subreactions mediating protein folding. Our findings illuminate the native conformational and functional chaperonin cycle directly within cells.
- Department of Cellular Biochemistry, Max Planck Institute of Biochemistry, Martinsried, Germany.
Organizational Affiliation: