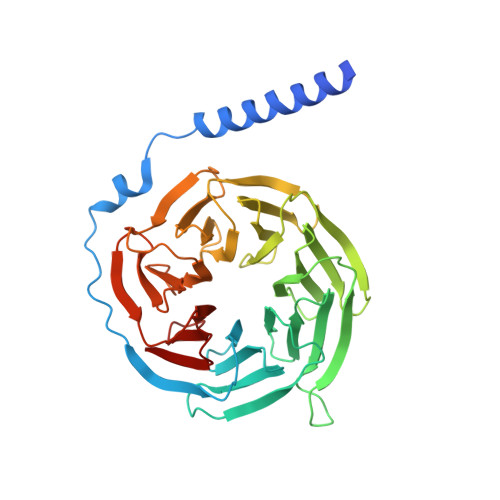
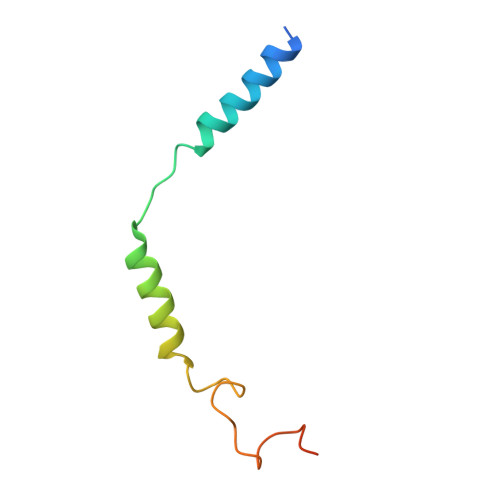
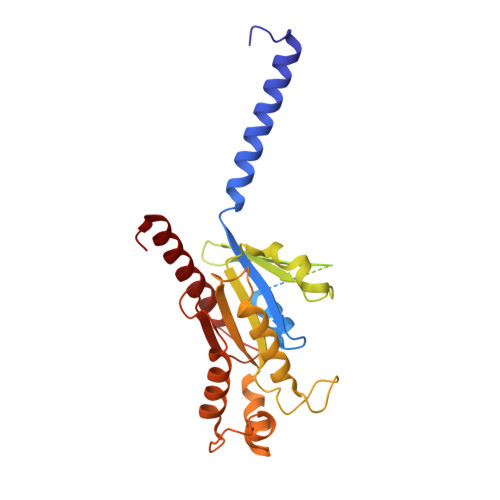
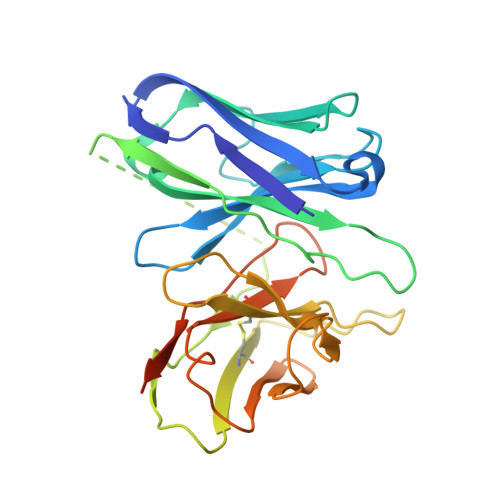
Orthosteric ligand selectivity and allosteric probe dependence at Hydroxycarboxylic acid receptor HCAR2.
Cheng, L., Sun, S., Wang, H., Zhao, C., Tian, X., Liu, Y., Fu, P., Shao, Z., Chai, R., Yan, W.(2023) Signal Transduct Target Ther 8: 364-364
- PubMed: 37743365 Search on PubMedSearch on PubMed Central
- DOI: https://doi.org/10.1038/s41392-023-01625-y
- Primary Citation Related Structures:
8JZ7 - PubMed Abstract:
Hydroxycarboxylic acid receptor 2 (HCAR2), a member of Class A G-protein-coupled receptor (GPCR) family, plays a pivotal role in anti-lipolytic and anti-inflammatory effects, establishing it as a significant therapeutic target for treating dyslipidemia and inflammatory diseases. However, the mechanism underlying the signaling of HCAR2 induced by various types of ligands remains elusive. In this study, we elucidate the cryo-electron microscopy (cryo-EM) structure of G i -coupled HCAR2 in complex with a selective agonist, MK-6892, resolved to a resolution of 2.60 Å. Our structural analysis reveals that MK-6892 occupies not only the orthosteric binding pocket (OBP) but also an extended binding pocket (EBP) within HCAR2. Pharmacological assays conducted in this study demonstrate that the OBP is a critical determinant for ligand selectivity among the HCARs subfamily. Moreover, we investigate the pharmacological properties of the allosteric modulator compound 9n, revealing its probe-dependent behavior on HCAR2 in response to varying orthosteric agonists. Collectively, our findings provide invaluable structural insights that contribute to a deeper understanding of the regulatory mechanisms governing HCAR2 signaling transduction mediated by both orthosteric and allosteric ligands.
- Department of Otolaryngology Head and Neck Surgery, Sichuan Provincial People's Hospital, University of Electronic Science and Technology of China, Chengdu, 610000, China.
Organizational Affiliation: