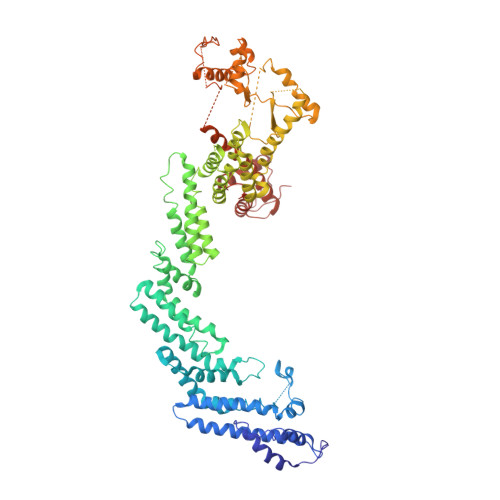
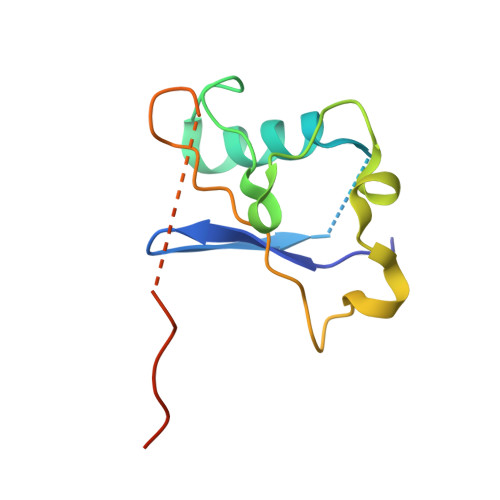
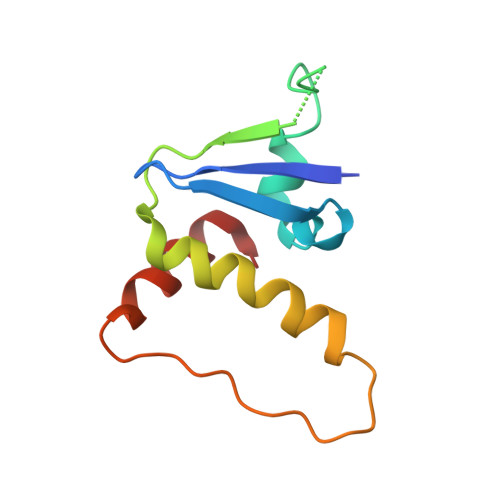
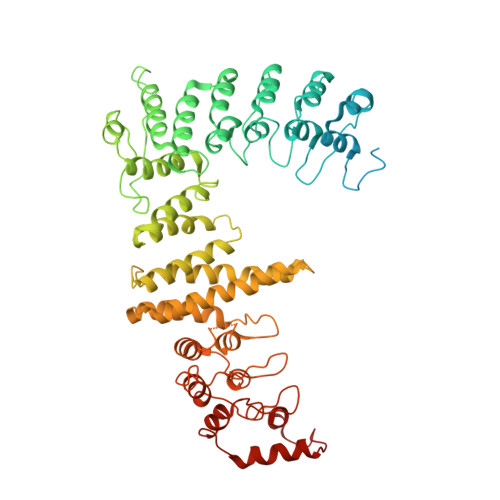
Structural insights into the ubiquitylation strategy of the oligomeric CRL2 FEM1B E3 ubiquitin ligase.
Dai, Z., Liang, L., Wang, W., Zuo, P., Yu, S., Liu, Y., Zhao, X., Lu, Y., Jin, Y., Zhang, F., Ding, D., Deng, W., Yin, Y.(2024) EMBO J 43: 1089-1109
- PubMed: 38360992 Search on PubMedSearch on PubMed Central
- DOI: https://doi.org/10.1038/s44318-024-00047-y
- Primary Citation Related Structures:
8IJ1, 8JE1, 8JE2 - PubMed Abstract:
Cullin-RING E3 ubiquitin ligase (CRL) family members play critical roles in numerous biological processes and diseases including cancer and Alzheimer's disease. Oligomerization of CRLs has been reported to be crucial for the regulation of their activities. However, the structural basis for its regulation and mechanism of its oligomerization are not fully known. Here, we present cryo-EM structures of oligomeric CRL2 FEM1B in its unneddylated state, neddylated state in complex with BEX2 as well as neddylated state in complex with FNIP1/FLCN. These structures reveal that asymmetric dimerization of N8-CRL2 FEM1B is critical for the ubiquitylation of BEX2 while FNIP1/FLCN is ubiquitylated by monomeric CRL2 FEM1B . Our data present an example of the asymmetric homo-dimerization of CRL. Taken together, this study sheds light on the ubiquitylation strategy of oligomeric CRL2 FEM1B according to substrates with different scales.
- Institute of Systems Biomedicine, Beijing Key Laboratory of Tumor Systems Biology, School of Basic Medical Sciences, Peking University Health Science Center, Beijing, 100191, China.
Organizational Affiliation: