Structure Of Human Lgi1-Adam22 Complex In Space Group C2
Liu, H.L., Lin, Z.H., Xu, F.To be published.
Experimental Data Snapshot
Starting Model: experimental
View more details
Macromolecule Content
Entity ID: 1 | |||||
|---|---|---|---|---|---|
| Molecule | Chains | Sequence Length | Organism | Details | Image |
| Disintegrin and metalloproteinase domain-containing protein 22 | 486 | Homo sapiens | Mutation(s): 0 Gene Names: ADAM22, MDC2 | 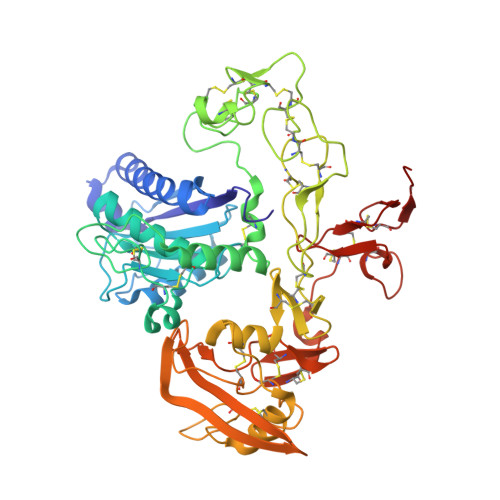 | |
UniProt & NIH Common Fund Data Resources | |||||
PHAROS: Q9P0K1 GTEx: ENSG00000008277 | |||||
Entity Groups | |||||
| Sequence Clusters | 30% Identity50% Identity70% Identity90% Identity95% Identity100% Identity | ||||
| UniProt Group | Q9P0K1 | ||||
Glycosylation | |||||
| Glycosylation Sites: 3 | Go to GlyGen: Q9P0K1-1 | ||||
Sequence AnnotationsExpand | |||||
Reference Sequence | |||||
Entity ID: 2 | |||||
|---|---|---|---|---|---|
| Molecule | Chains | Sequence Length | Organism | Details | Image |
| Leucine-rich glioma-inactivated protein 1 | 526 | Homo sapiens | Mutation(s): 0 Gene Names: LGI1, EPT, UNQ775/PRO1569 | 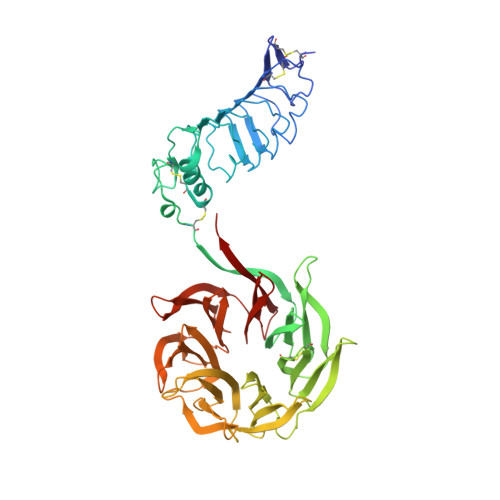 | |
UniProt & NIH Common Fund Data Resources | |||||
PHAROS: O95970 GTEx: ENSG00000108231 | |||||
Entity Groups | |||||
| Sequence Clusters | 30% Identity50% Identity70% Identity90% Identity95% Identity100% Identity | ||||
| UniProt Group | O95970 | ||||
Glycosylation | |||||
| Glycosylation Sites: 3 | Go to GlyGen: O95970-1 | ||||
Sequence AnnotationsExpand | |||||
Reference Sequence | |||||
| Ligands 2 Unique | |||||
|---|---|---|---|---|---|
| ID | Chains | Name / Formula / InChI Key | 2D Diagram | 3D Interactions | |
| NAG Download:Ideal Coordinates CCD File | AA [auth D] CA [auth E] DA [auth E] EA [auth E] G [auth A] | 2-acetamido-2-deoxy-beta-D-glucopyranose C8 H15 N O6 OVRNDRQMDRJTHS-FMDGEEDCSA-N |  | ||
| CA Download:Ideal Coordinates CCD File | BA [auth D] FA [auth E] J [auth A] JA [auth F] K [auth A] | CALCIUM ION Ca BHPQYMZQTOCNFJ-UHFFFAOYSA-N |  | ||
| Length ( Å ) | Angle ( ˚ ) |
|---|---|
| a = 373.503 | α = 90 |
| b = 232.369 | β = 94.97 |
| c = 80.035 | γ = 90 |
| Software Name | Purpose |
|---|---|
| HKL-2000 | data reduction |
| HKL-2000 | data scaling |
| PHASER | phasing |
| PHENIX | refinement |
| Funding Organization | Location | Grant Number |
|---|---|---|
| National Natural Science Foundation of China (NSFC) | China | 31270767 |