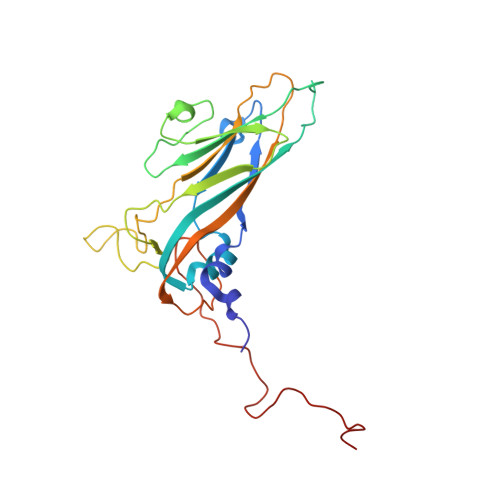
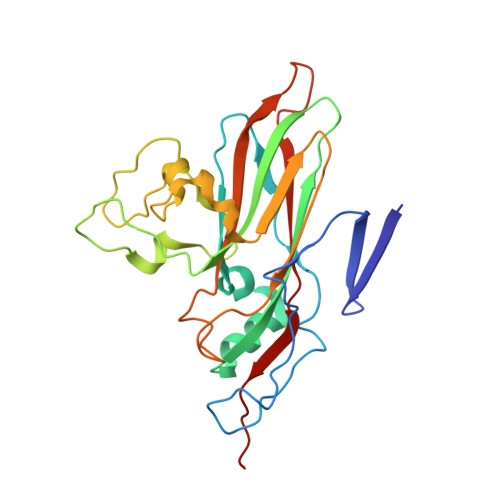
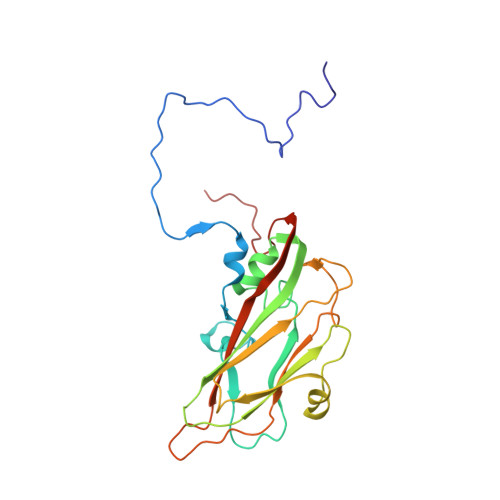
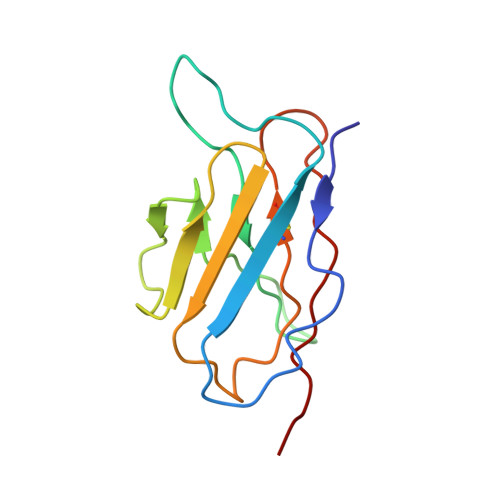
Structural Basis for the Immunogenicity of the C-Terminus of VP1 of Echovirus 3 Revealed by the Binding of a Neutralizing Antibody.
Qi, S., Fu, W., Fan, J., Zhang, L., Zheng, B., Wang, K., Wang, X., Zhu, L., Li, X., Zhang, Y.(2022) Viruses 14
- PubMed: 36366420
- DOI: https://doi.org/10.3390/v14112322
- Primary Citation Related Structures:
8GSC, 8GSD, 8GSE, 8GSF - PubMed Abstract:
Echovirus 3 (E3), a serotype of human enterovirus B (HEV-B), causes severe diseases in infants. Here, we determined the structures of E3 with a monoclonal antibody (MAb) 6D10 by cryo-EM to comprehensively understand the specificities and the immunological characteristic of this serotype. The solved cryo-EM structures of the F-, A-, and E-particles of E3 bound with 6D10 revealed the structural features of the virus-antibody interface. Importantly, the structures of E-particles bound with 6D10 revealed for the first time the nature of the C-terminus of VP1 for HEV-Bs at the structural level. The highly immunogenic nature of this region in the E-particles provides new strategies for vaccine development for HEV-Bs.
- CAS Key Laboratory of Infection and Immunity, National Laboratory of Macromolecules, Institute of Biophysics, Chinese Academy of Sciences, Beijing 100101, China.
Organizational Affiliation: