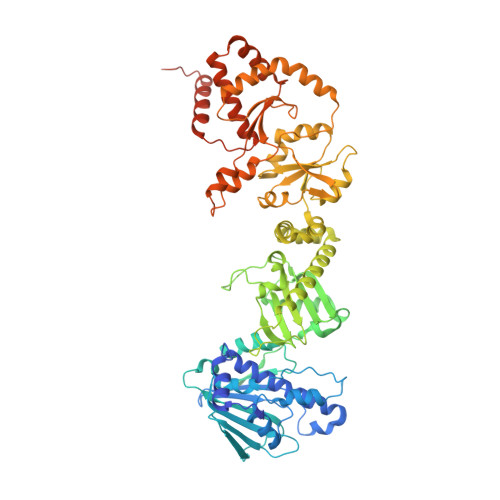
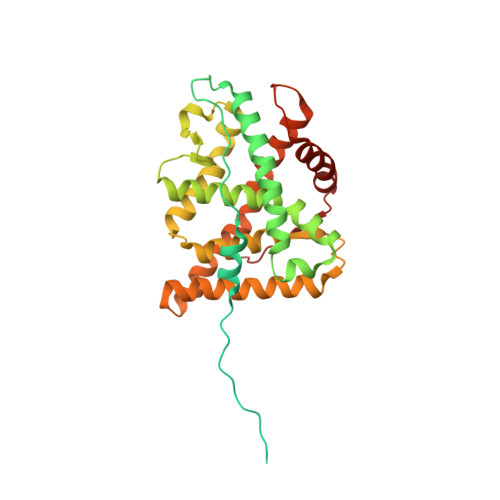
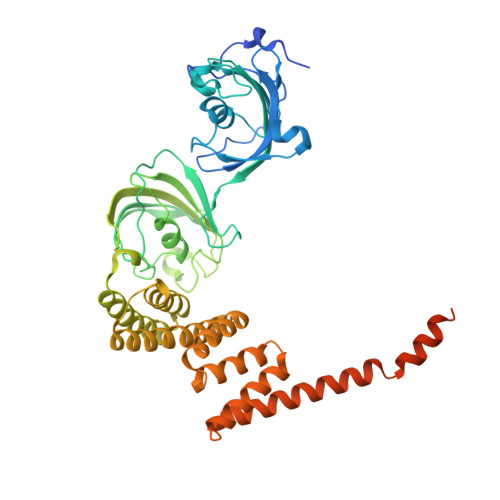
Cryo-EM reveals how Hsp90 and FKBP immunophilins co-regulate the glucocorticoid receptor.
Noddings, C.M., Johnson, J.L., Agard, D.A.(2023) Nat Struct Mol Biol 30: 1867-1877
- PubMed: 37945740 Search on PubMedSearch on PubMed Central
- DOI: https://doi.org/10.1038/s41594-023-01128-y
- Primary Citation Related Structures:
8FFV, 8FFW - PubMed Abstract:
Hsp90 is an essential molecular chaperone responsible for the folding and activation of hundreds of 'client' proteins, including the glucocorticoid receptor (GR). Previously, we revealed that Hsp70 and Hsp90 remodel the conformation of GR to regulate ligand binding, aided by co-chaperones. In vivo, the co-chaperones FKBP51 and FKBP52 antagonistically regulate GR activity, but a molecular understanding is lacking. Here we present a 3.01 Å cryogenic electron microscopy structure of the human GR:Hsp90:FKBP52 complex, revealing how FKBP52 integrates into the GR chaperone cycle and directly binds to the active client, potentiating GR activity in vitro and in vivo. We also present a 3.23 Å cryogenic electron microscopy structure of the human GR:Hsp90:FKBP51 complex, revealing how FKBP51 competes with FKBP52 for GR:Hsp90 binding and demonstrating how FKBP51 can act as a potent antagonist to FKBP52. Altogether, we demonstrate how FKBP51 and FKBP52 integrate into the GR chaperone cycle to advance GR to the next stage of maturation.
- Department of Biochemistry and Biophysics, University of California, San Francisco, San Francisco, CA, USA.
Organizational Affiliation: