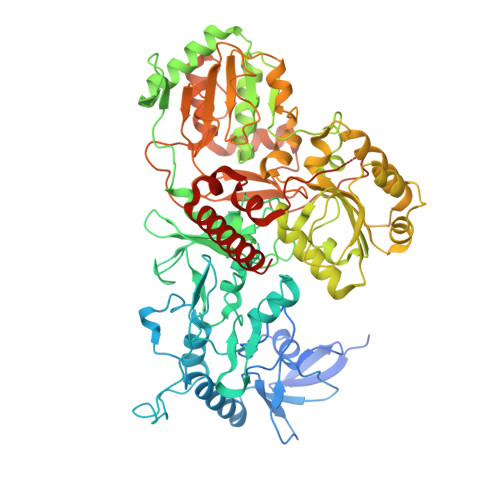
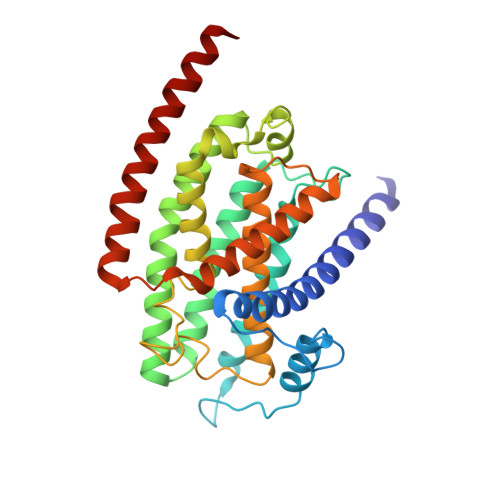
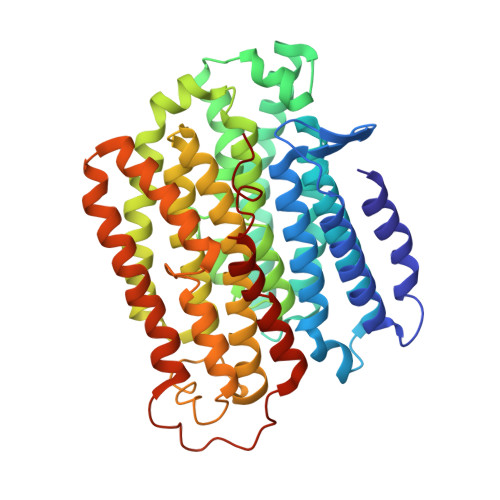
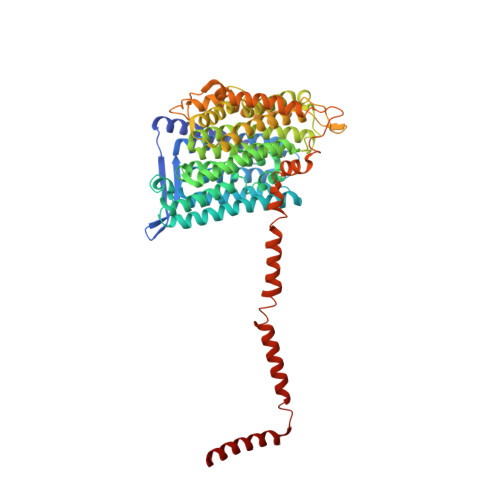
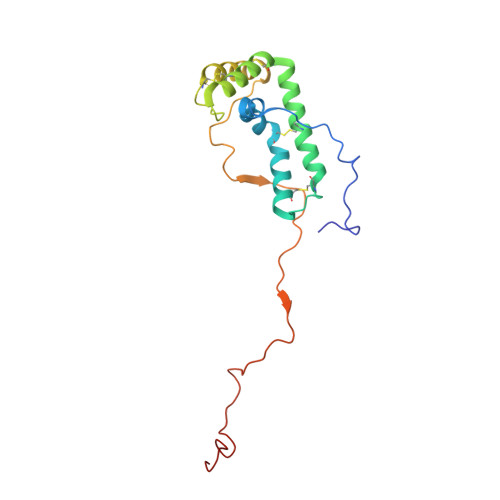
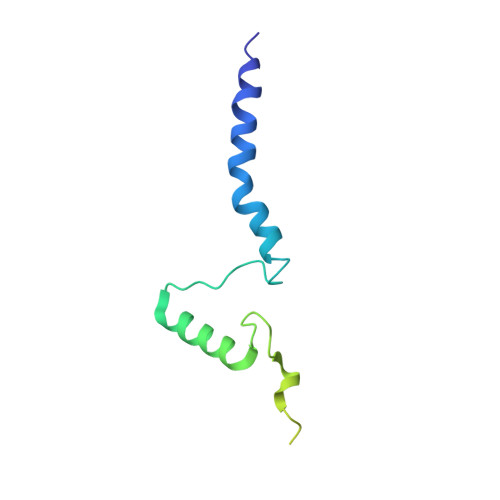
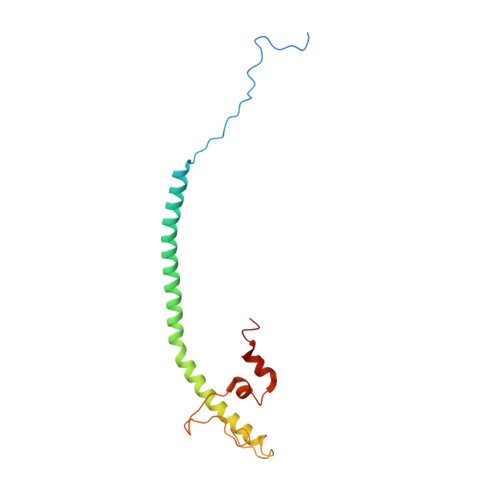
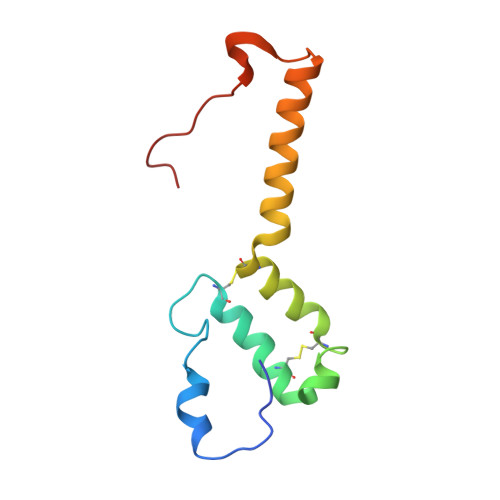
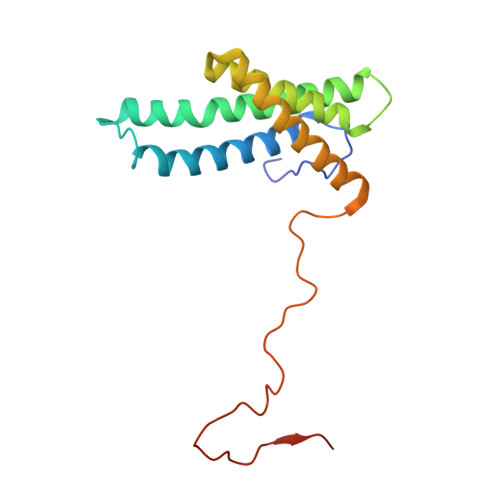
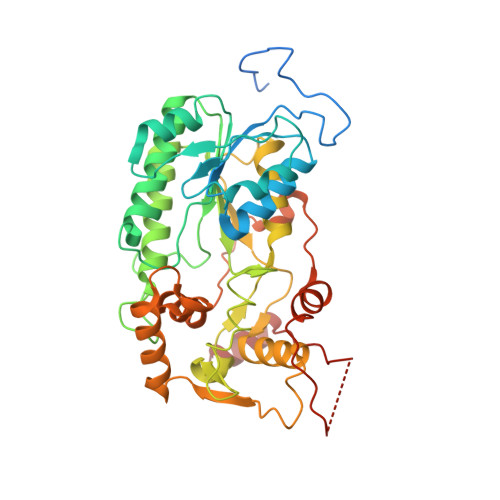
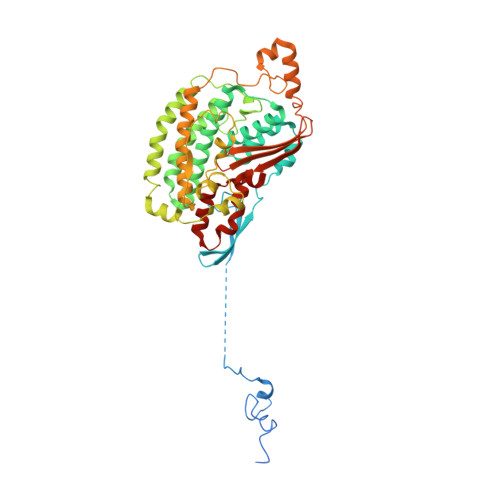
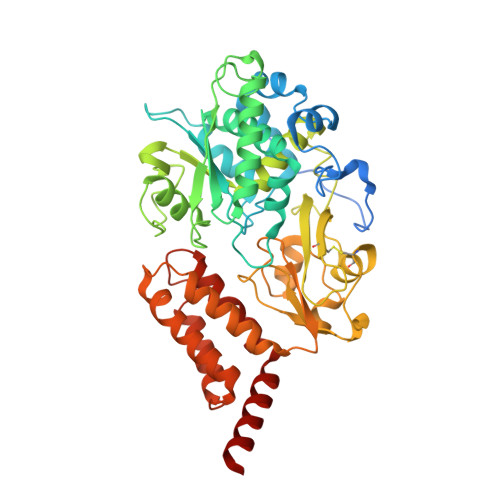
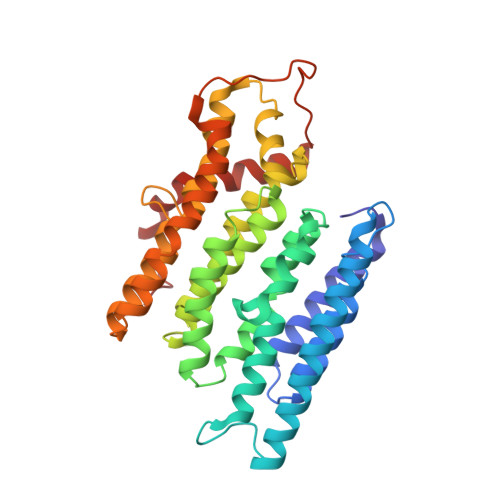
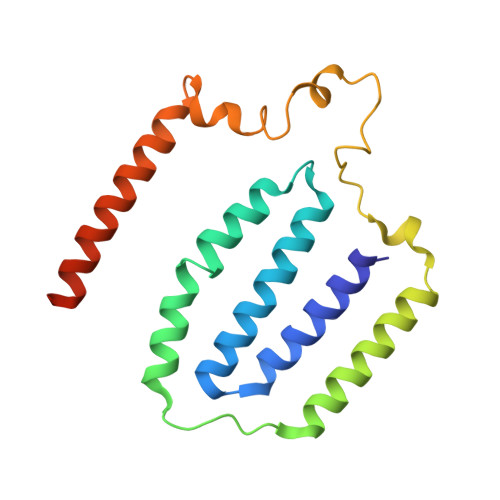
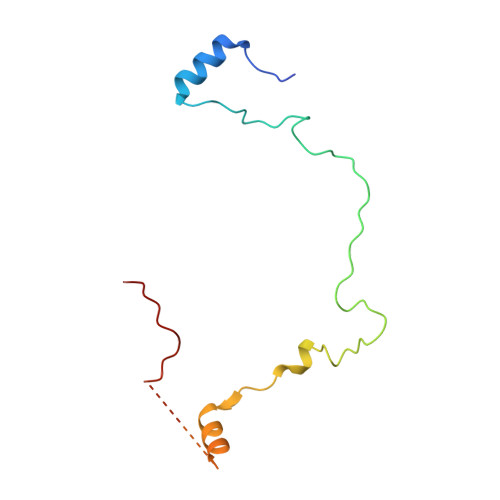
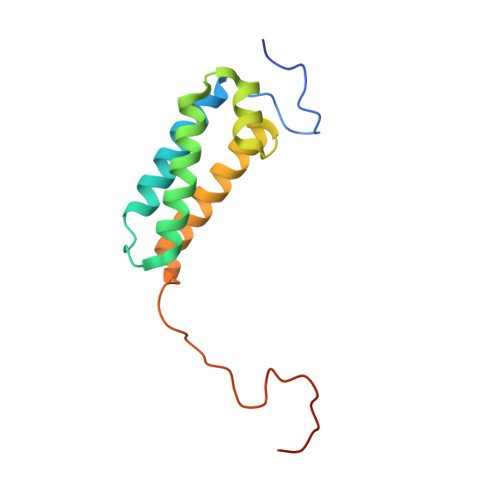
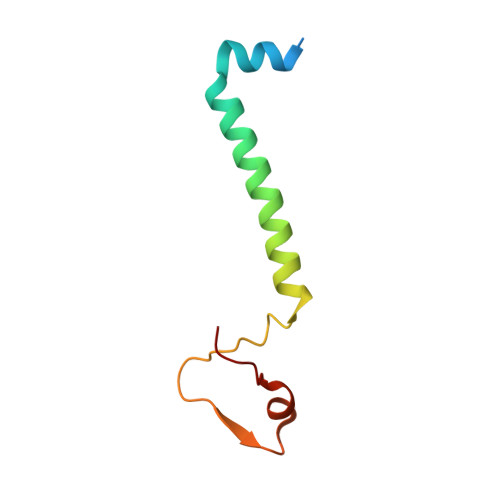
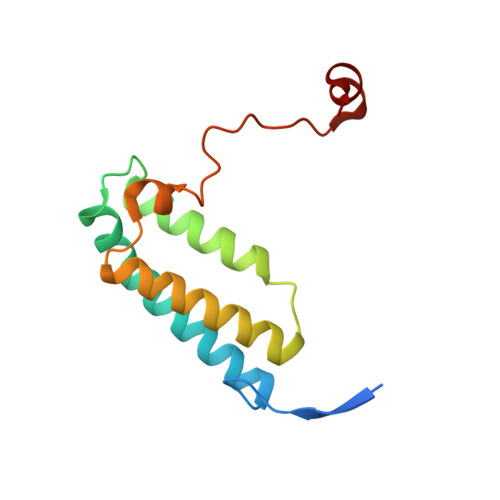
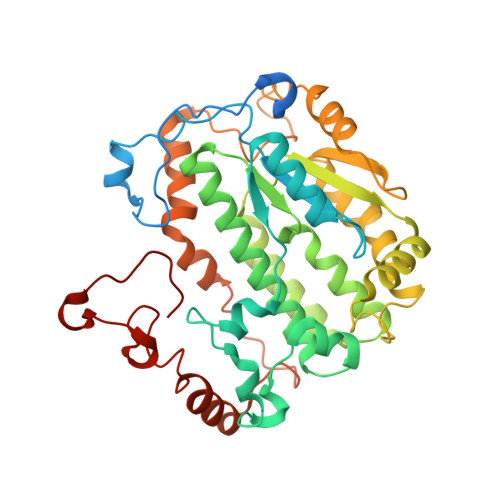
Resting mitochondrial complex I from Drosophila melanogaster adopts a helix-locked state.
Padavannil, A., Murari, A., Rhooms, S.K., Owusu-Ansah, E., Letts, J.A.(2023) Elife 12
- PubMed: 36952377 Search on PubMedSearch on PubMed Central
- DOI: https://doi.org/10.7554/eLife.84415
- Primary Citation Related Structures:
8ESW, 8ESZ - PubMed Abstract:
Respiratory complex I is a proton-pumping oxidoreductase key to bioenergetic metabolism. Biochemical studies have found a divide in the behavior of complex I in metazoans that aligns with the evolutionary split between Protostomia and Deuterostomia. Complex I from Deuterostomia including mammals can adopt a biochemically defined off-pathway 'deactive' state, whereas complex I from Protostomia cannot. The presence of off-pathway states complicates the interpretation of structural results and has led to considerable mechanistic debate. Here, we report the structure of mitochondrial complex I from the thoracic muscles of the model protostome Drosophila melanogaster . We show that although D. melanogaster complex I ( Dm -CI) does not have a NEM-sensitive deactive state, it does show slow activation kinetics indicative of an off-pathway resting state. The resting-state structure of Dm -CI from the thoracic muscle reveals multiple conformations. We identify a helix-locked state in which an N-terminal α-helix on the NDUFS4 subunit wedges between the peripheral and membrane arms. Comparison of the Dm -CI structure and conformational states to those observed in bacteria, yeast, and mammals provides insight into the roles of subunits across organisms, explains why the Dm -CI off-pathway resting state is NEM insensitive, and raises questions regarding current mechanistic models of complex I turnover.
- Department of Molecular and Cellular Biology, University of California, Davis, Davis, United States.
Organizational Affiliation: