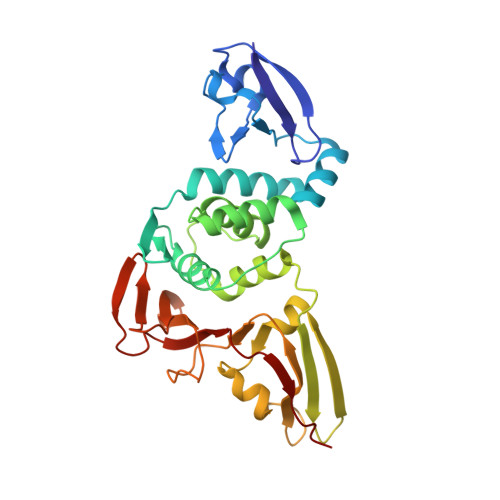
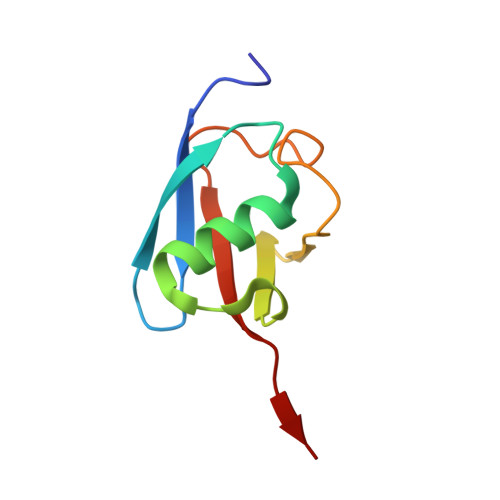
The substrate selectivity of papain-like proteases from human-infecting coronaviruses correlates with innate immune suppression.
Xiong, Y., Huang, B., Yang, Y., Fu, X., Fu, Z., Xu, H., Liu, M., Cao, D., Zhang, M., Yang, H., Niu, X., Yu, C., Huang, H.(2023) Sci Signal 16: eade1985-eade1985
- PubMed: 37130166 Search on PubMed
- DOI: https://doi.org/10.1126/scisignal.ade1985
- Primary Citation Related Structures:
7WFC - PubMed Abstract:
Coronaviruses that can infect humans can cause either common colds (HCoV-NL63, HCoV-229E, HCoV-HKU1, and HCoV-OC43) or severe respiratory symptoms (SARS-CoV-2, SARS-CoV, and MERS-CoV). The papain-like proteases (PLPs) of SARS-CoV, SARS-CoV-2, MERS-CoV, and HCoV-NL63 function in viral innate immune evasion and have deubiquitinating (DUB) and deISGylating activities. We identified the PLPs of HCoV-229E, HCoV-HKU1, and HCoV-OC43 and found that their enzymatic properties correlated with their ability to suppress innate immune responses. A conserved noncatalytic aspartic acid residue was critical for both DUB and deISGylating activities, but the PLPs had differing ubiquitin (Ub) chain cleavage selectivities and binding affinities for Ub, K48-linked diUb, and interferon-stimulated gene 15 (ISG15) substrates. The crystal structure of HKU1-PLP2 in complex with Ub revealed binding interfaces that accounted for the unusually high binding affinity between this PLP and Ub. In cellular assays, the PLPs from the severe disease-causing coronaviruses strongly suppressed innate immune IFN-I and NF-κB signaling and stimulated autophagy, whereas the PLPs from the mild disease-causing coronaviruses generally showed weaker effects on immune suppression and autophagy induction. In addition, a PLP from a SARS-CoV-2 variant of concern showed increased suppression of innate immune signaling pathways. Overall, these results demonstrated that the DUB and deISGylating activities and substrate selectivities of these PLPs differentially contribute to viral innate immune evasion and may affect viral pathogenicity.
- State Key Laboratory of Chemical Oncogenomics, Guangdong Provincial Key Laboratory of Chemical Genomics, Laboratory of Structural Biology and Drug Discovery, Laboratory of Ubiquitination and Targeted Therapy, School of Chemical Biology and Biotechnology, Peking University Shenzhen Graduate School, Shenzhen, Guangdong 518055, China.
Organizational Affiliation: