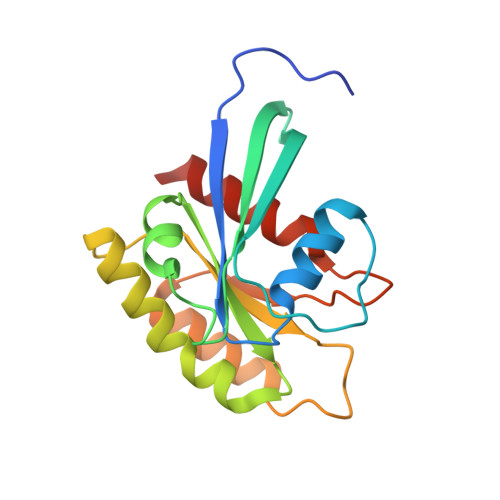
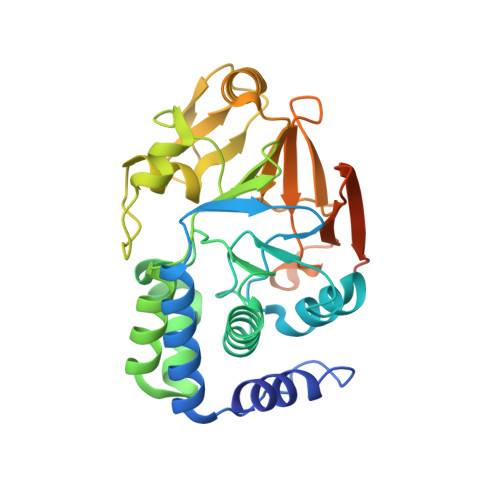
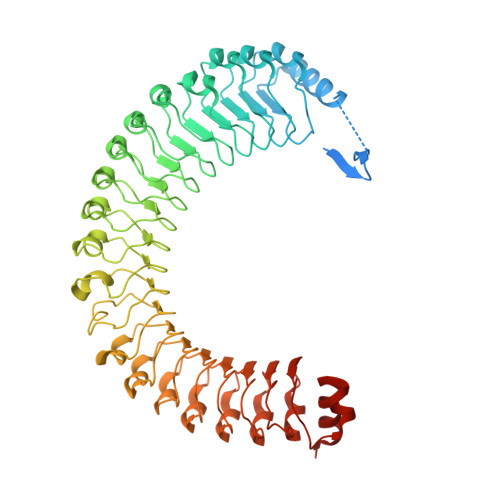
Structure-function analysis of the SHOC2-MRAS-PP1C holophosphatase complex.
Kwon, J.J., Hajian, B., Bian, Y., Young, L.C., Amor, A.J., Fuller, J.R., Fraley, C.V., Sykes, A.M., So, J., Pan, J., Baker, L., Lee, S.J., Wheeler, D.B., Mayhew, D.L., Persky, N.S., Yang, X., Root, D.E., Barsotti, A.M., Stamford, A.W., Perry, C.K., Burgin, A., McCormick, F., Lemke, C.T., Hahn, W.C., Aguirre, A.J.(2022) Nature 609: 408-415
- PubMed: 35831509 Search on PubMedSearch on PubMed Central
- DOI: https://doi.org/10.1038/s41586-022-04928-2
- Primary Citation Related Structures:
7T7A, 7UPI - PubMed Abstract:
Receptor tyrosine kinase (RTK)-RAS signalling through the downstream mitogen-activated protein kinase (MAPK) cascade regulates cell proliferation and survival. The SHOC2-MRAS-PP1C holophosphatase complex functions as a key regulator of RTK-RAS signalling by removing an inhibitory phosphorylation event on the RAF family of proteins to potentiate MAPK signalling 1 . SHOC2 forms a ternary complex with MRAS and PP1C, and human germline gain-of-function mutations in this complex result in congenital RASopathy syndromes 2-5 . However, the structure and assembly of this complex are poorly understood. Here we use cryo-electron microscopy to resolve the structure of the SHOC2-MRAS-PP1C complex. We define the biophysical principles of holoenzyme interactions, elucidate the assembly order of the complex, and systematically interrogate the functional consequence of nearly all of the possible missense variants of SHOC2 through deep mutational scanning. We show that SHOC2 binds PP1C and MRAS through the concave surface of the leucine-rich repeat region and further engages PP1C through the N-terminal disordered region that contains a cryptic RVXF motif. Complex formation is initially mediated by interactions between SHOC2 and PP1C and is stabilized by the binding of GTP-loaded MRAS. These observations explain how mutant versions of SHOC2 in RASopathies and cancer stabilize the interactions of complex members to enhance holophosphatase activity. Together, this integrative structure-function model comprehensively defines key binding interactions within the SHOC2-MRAS-PP1C holophosphatase complex and will inform therapeutic development .
- Cancer Program, Broad Institute of MIT and Harvard, Cambridge, MA, USA.
Organizational Affiliation: