Crystal structure of R14A-R20A human Galectin-7 mutant in presence of lactose
Pham, N.T.H., Calmettes, C., Doucet, N.To be published.
Experimental Data Snapshot
Starting Model: experimental
View more details
wwPDB Validation 3D Report Full Report
Macromolecule Content
Entity ID: 1 | |||||
|---|---|---|---|---|---|
| Molecule | Chains | Sequence Length | Organism | Details | Image |
| Galectin-7 | 135 | Homo sapiens | Mutation(s): 2 Gene Names: LGALS7, PIG1, LGALS7B | 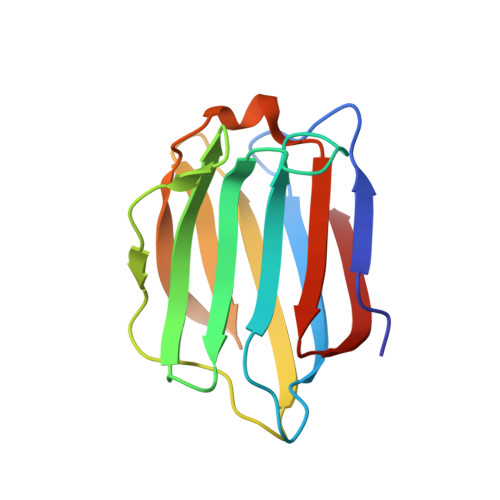 | |
UniProt & NIH Common Fund Data Resources | |||||
PHAROS: P47929 | |||||
Entity Groups | |||||
| Sequence Clusters | 30% Identity50% Identity70% Identity90% Identity95% Identity100% Identity | ||||
| UniProt Group | P47929 | ||||
Sequence AnnotationsExpand | |||||
Reference Sequence | |||||
| Ligands 2 Unique | |||||
|---|---|---|---|---|---|
| ID | Chains | Name / Formula / InChI Key | 2D Diagram | 3D Interactions | |
| TRS Download:Ideal Coordinates CCD File | E [auth A] | 2-AMINO-2-HYDROXYMETHYL-PROPANE-1,3-DIOL C4 H12 N O3 LENZDBCJOHFCAS-UHFFFAOYSA-O |  | ||
| EDO Download:Ideal Coordinates CCD File | D [auth A] | 1,2-ETHANEDIOL C2 H6 O2 LYCAIKOWRPUZTN-UHFFFAOYSA-N |  | ||
Entity ID: 2 | |||||
|---|---|---|---|---|---|
| ID | Chains | Name | Type/Class | 2D Diagram | 3D Interactions |
| PRD_900004 Query on PRD_900004 | C | beta-lactose | Oligosaccharide / Nutrient |  |
| Length ( Å ) | Angle ( ˚ ) |
|---|---|
| a = 53.97 | α = 90 |
| b = 66.16 | β = 90 |
| c = 70.55 | γ = 90 |
| Software Name | Purpose |
|---|---|
| PHENIX | refinement |
| XDS | data reduction |
| XSCALE | data scaling |
| PHASER | phasing |
| PDB_EXTRACT | data extraction |
| Funding Organization | Location | Grant Number |
|---|---|---|
| Natural Sciences and Engineering Research Council (NSERC, Canada) | Canada | RGPIN 2016-05557 |
| National Institutes of Health/National Institute of General Medical Sciences (NIH/NIGMS) | United States | R01GM105978 |
| Fonds de Recherche du Quebec - Sante (FRQS) | Canada | FRQ-S Research Scholar Senior Career Award (281993) |
| Fonds de Recherche du Quebec - Sante (FRQS) | Canada | FRQ-S Junior 1 (251848) |
| Natural Sciences and Engineering Research Council (NSERC, Canada) | Canada | RGPIN-2017-06091 |
| Fonds de Recherche du Quebec - Sante (FRQS) | Canada | FRQ-S Doctoral Training scholarship (287239) |