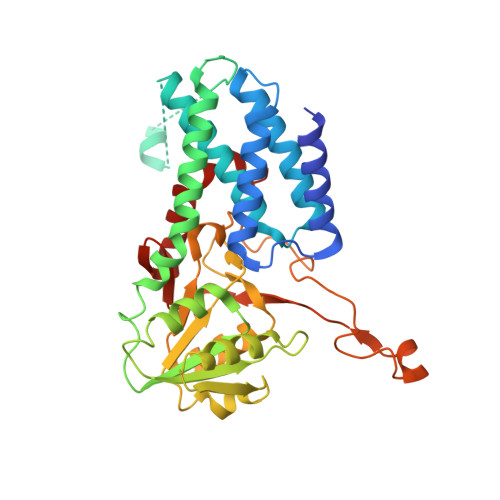
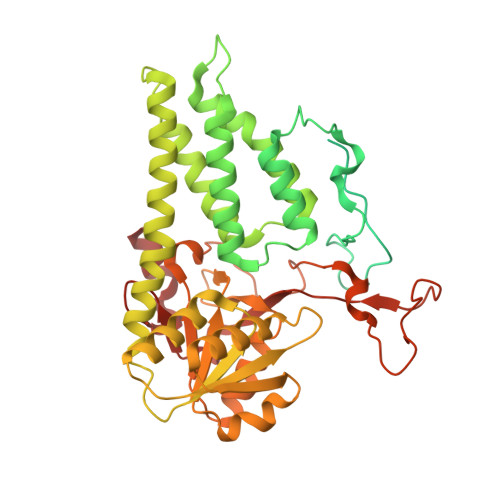
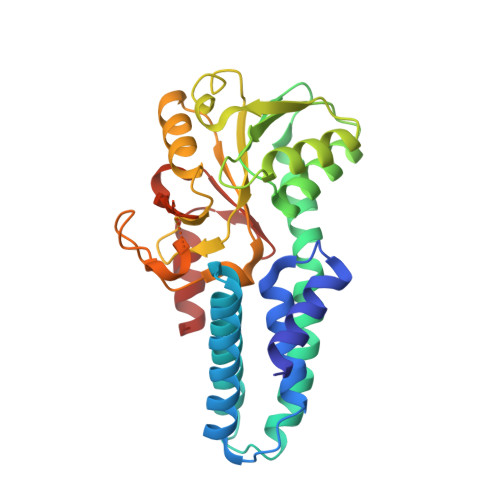
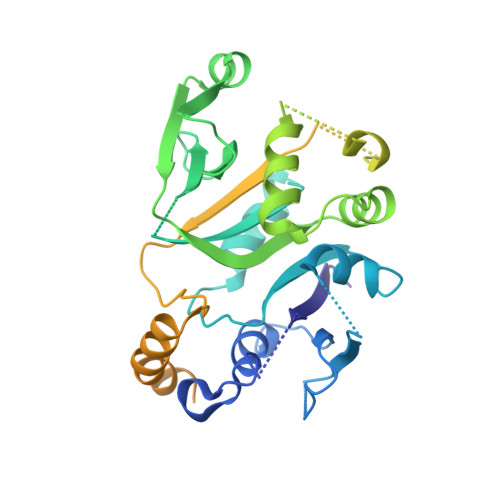
A point mutation in the nucleotide exchange factor eIF2B constitutively activates the integrated stress response by allosteric modulation.
Boone, M., Wang, L., Lawrence, R., Frost, A., Walter, P., Schoof, M.(2022) Elife 11
- PubMed: 35416150 Search on PubMedSearch on PubMed Central
- DOI: https://doi.org/10.7554/eLife.76171
- Primary Citation Related Structures:
7TRJ - PubMed Abstract:
In eukaryotic cells, stressors reprogram the cellular proteome by activating the integrated stress response (ISR). In its canonical form, stress-sensing kinases phosphorylate the eukaryotic translation initiation factor eIF2 (eIF2-P), which ultimately leads to reduced levels of ternary complex required for initiation of mRNA translation. Previously we showed that translational control is primarily exerted through a conformational switch in eIF2's nucleotide exchange factor, eIF2B, which shifts from its active A-State conformation to its inhibited I-State conformation upon eIF2-P binding, resulting in reduced nucleotide exchange on eIF2 (Schoof et al. 2021). Here, we show functionally and structurally how a single histidine to aspartate point mutation in eIF2B's β subunit (H160D) mimics the effects of eIF2-P binding by promoting an I-State like conformation, resulting in eIF2-P independent activation of the ISR. These findings corroborate our previously proposed A/I-State model of allosteric ISR regulation.
- Howard Hughes Medical Institute, University of California at San Francisco, San Francisco, United States.
Organizational Affiliation: