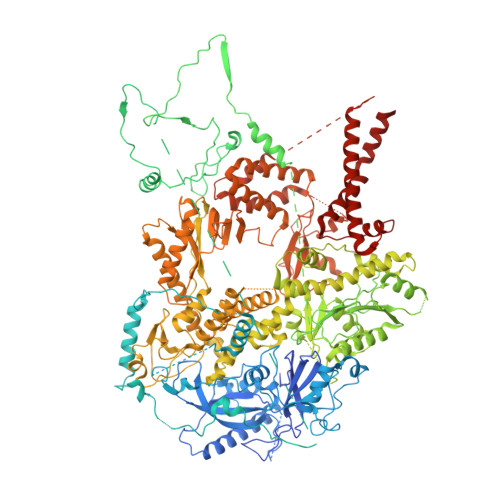
Cryo-EM structure of translesion DNA synthesis polymerase zeta with a base pair mismatch.
Malik, R., Johnson, R.E., Prakash, L., Prakash, S., Ubarretxena-Belandia, I., Aggarwal, A.K.(2022) Nat Commun 13: 1050-1050
- PubMed: 35217661
- DOI: https://doi.org/10.1038/s41467-022-28644-7
- Primary Citation of Related Structures:
7S0T - PubMed Abstract:
The B-family multi-subunit DNA polymerase ζ (Polζ) is important for translesion DNA synthesis (TLS) during replication, due to its ability to extend synthesis past nucleotides opposite DNA lesions and mismatched base pairs. We present a cryo-EM structure of Saccharomyces cerevisiae Polζ with an A:C mismatch at the primer terminus. The structure shows how the Polζ active site responds to the mismatched duplex DNA distortion, including the loosening of key protein-DNA interactions and a fingers domain in an "open" conformation, while the incoming dCTP is still able to bind for the extension reaction. The structure of the mismatched DNA-Polζ ternary complex reveals insights into mechanisms that either stall or favor continued DNA synthesis in eukaryotes.
- Department of Pharmacological Sciences, Icahn School of Medicine at Mount Sinai, New York, New York, NY, USA.
Organizational Affiliation: