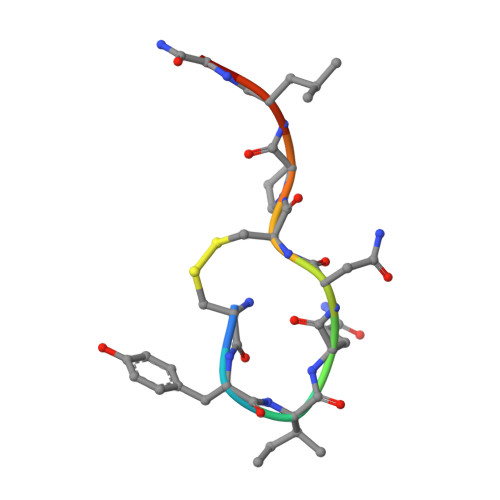
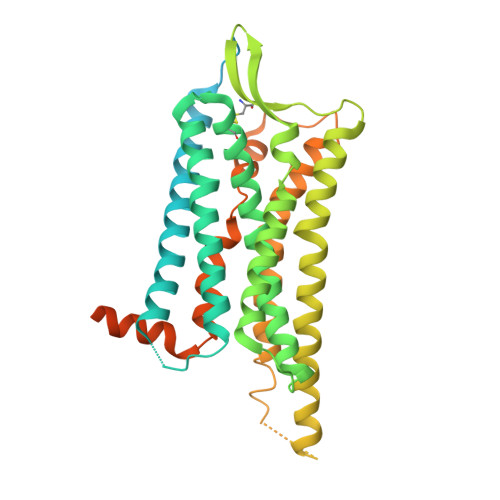
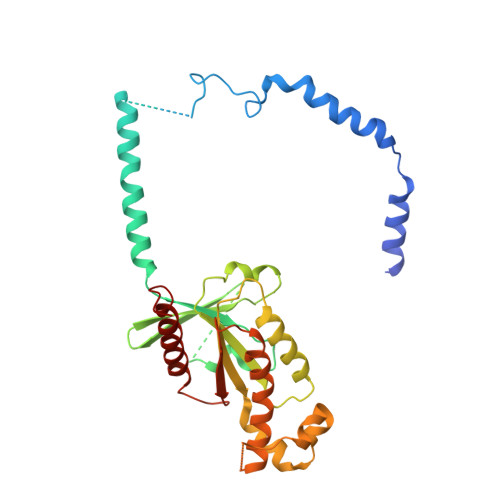
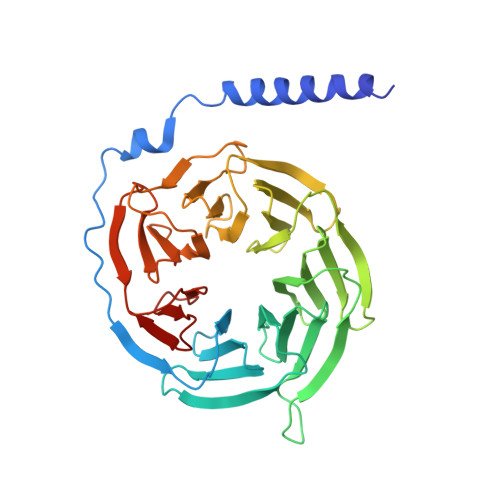
The oxytocin signaling complex reveals a molecular switch for cation dependence.
Meyerowitz, J.G., Robertson, M.J., Barros-Alvarez, X., Panova, O., Nwokonko, R.M., Gao, Y., Skiniotis, G.(2022) Nat Struct Mol Biol 29: 274-281
- PubMed: 35241813
- DOI: https://doi.org/10.1038/s41594-022-00728-4
- Primary Citation Related Structures:
7RYC - PubMed Abstract:
Oxytocin (OT) and vasopressin (AVP) are conserved peptide signaling hormones that are critical for diverse processes including osmotic homeostasis, reproduction, lactation and social interaction. OT acts through the oxytocin receptor (OTR), a magnesium-dependent G protein-coupled receptor that is a therapeutic target for treatment of postpartum hemorrhage, dysfunctional labor and autism. However, the molecular mechanisms that underlie OTR activation by OT and the dependence on magnesium remain unknown. Here we present the wild-type active-state structure of human OTR bound to OT and miniG q/i determined by cryo-EM. The structure reveals a unique activation mechanism adopted by OTR involving both the formation of a Mg 2+ coordination complex between OT and the receptor, and disruption of transmembrane helix 7 (TM7) by OT. Our functional assays demonstrate the role of TM7 disruption and provide the mechanism of full agonism by OT and partial agonism by OT analogs. Furthermore, we find that the identity of a single cation-coordinating residue across vasopressin family receptors determines whether the receptor is cation-dependent. Collectively, these results demonstrate how the Mg 2+ -dependent OTR is activated by OT, provide essential information for structure-based drug discovery efforts and shed light on the molecular determinants of cation dependence of vasopressin family receptors throughout the animal kingdom.
- Department of Molecular and Cellular Physiology, Stanford University School of Medicine, Stanford, CA, USA.
Organizational Affiliation: