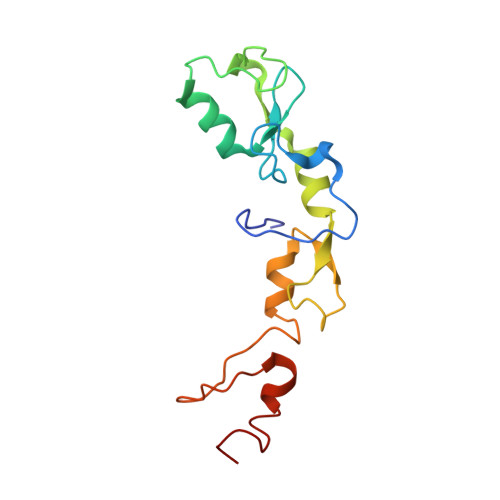
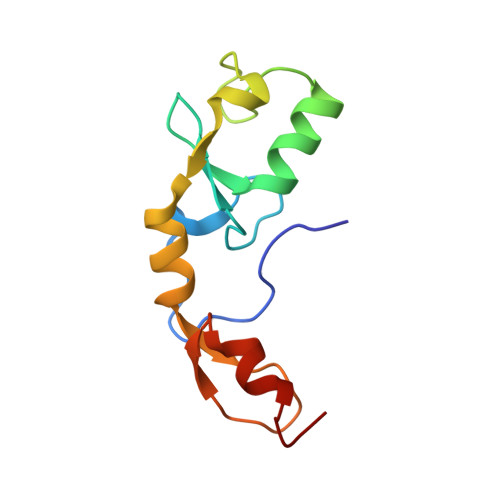
The structure and ubiquitin binding properties of TRAF RING heterodimers.
Das, A., Middleton, A.J., Padala, P., Ledgerwood, E.C., Mace, P.D., Day, C.L.(2021) J Mol Biology : 166844-166844
- PubMed: 33539883 Search on PubMed
- DOI: https://doi.org/10.1016/j.jmb.2021.166844
- Primary Citation Related Structures:
7L3L - PubMed Abstract:
Tumour necrosis factor (TNF) receptor associated factor (TRAF) family members share a common domain architecture, but play non-redundant physiological roles in cell signalling. At the N terminus, most TRAFs have a RING domain, followed by a series of Zinc finger (ZF) domains. The RING domain of TRAF6 dimerizes, and the RING homodimer together with the first ZF assembles ubiquitin chains that form a platform which facilitates activation of downstream kinases. The RING dimer interface is conserved amongst TRAF proteins, suggesting that functional heterodimers could be possible. Here we report the structure of the TRAF5-TRAF6 RING heterodimer, which accounts for the stability of the heterodimer as well as its ability to assemble ubiquitin chains. We also show that the RING domain of TRAF6 heterodimerizes with TRAF3 and TRAF2, and demonstrate that the linker helix and first ZF of TRAF2 can cooperate with TRAF6 to promote chain assembly. Collectively our results suggest that TRAF RING homo- and hetero-dimers have the potential to bridge interaction of nearby TRAF trimers and modulate TRAF-mediated signalling.
- Department of Biochemistry, School of Biomedical Sciences, University of Otago, Dunedin 9054, New Zealand.
Organizational Affiliation: