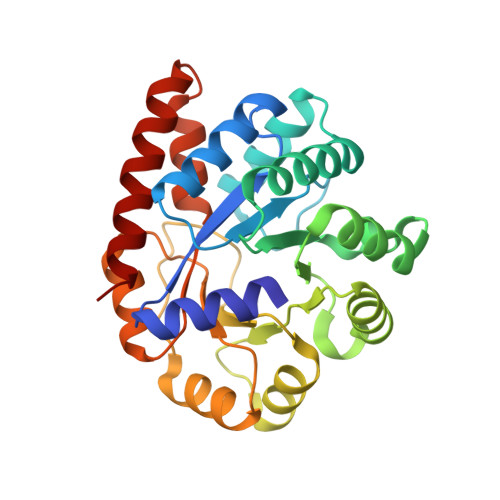
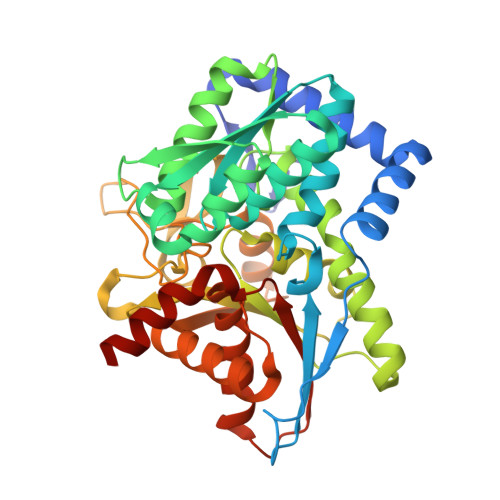
The aminoacrylate form of the wild-type Salmonella typhimurium Tryptophan Synthase in complex with inhibitor N-(4'-trifluoromethoxybenzenesulfonyl)-2-amino-1-ethylphosphate (F9F) at the enzyme alpha-site, cesium ion at the metal coordination site and benzimidazole (BZI) at the enzyme beta-site at 1.35 Angstrom resolution.
Hilario, E., Dunn, M.F., Mueller, L.J.To be published.