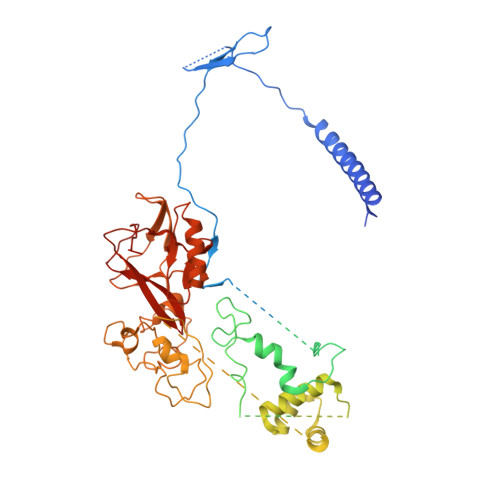
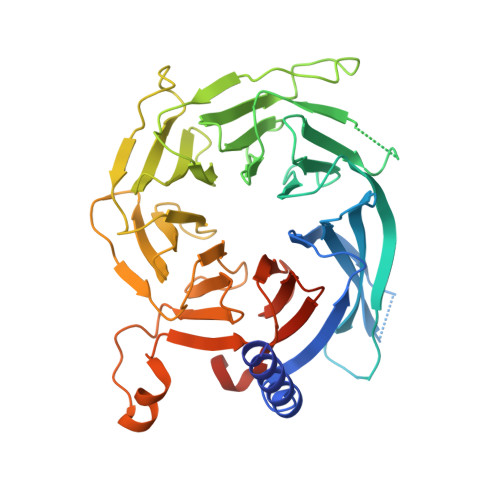
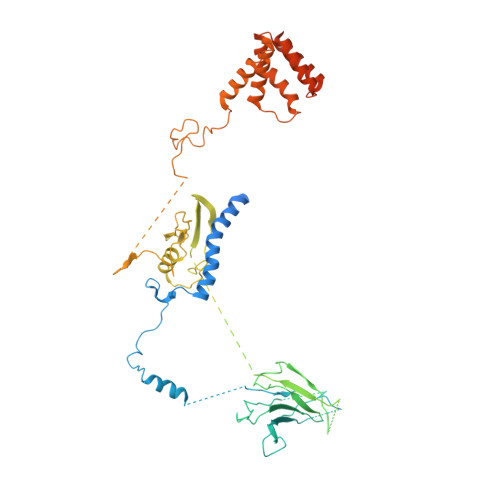
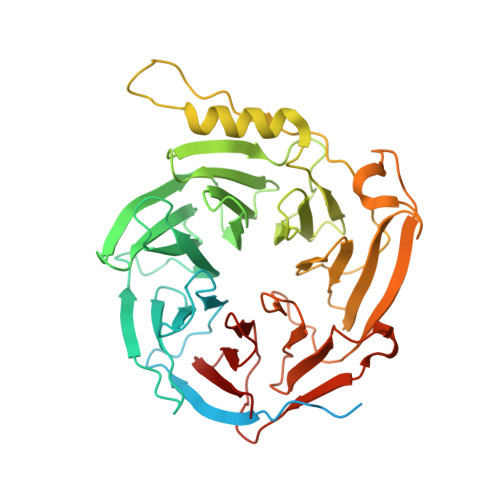
Structures of monomeric and dimeric PRC2:EZH1 reveal flexible modules involved in chromatin compaction.
Grau, D., Zhang, Y., Lee, C.H., Valencia-Sanchez, M., Zhang, J., Wang, M., Holder, M., Svetlov, V., Tan, D., Nudler, E., Reinberg, D., Walz, T., Armache, K.J.(2021) Nat Commun 12: 714-714
- PubMed: 33514705 Search on PubMedSearch on PubMed Central
- DOI: https://doi.org/10.1038/s41467-020-20775-z
- Primary Citation Related Structures:
7KSO, 7KSR, 7KTP, 7KTQ - PubMed Abstract:
Polycomb repressive complex 2 (PRC2) is a histone methyltransferase critical for maintaining gene silencing during eukaryotic development. In mammals, PRC2 activity is regulated in part by the selective incorporation of one of two paralogs of the catalytic subunit, EZH1 or EZH2. Each of these enzymes has specialized biological functions that may be partially explained by differences in the multivalent interactions they mediate with chromatin. Here, we present two cryo-EM structures of PRC2:EZH1, one as a monomer and a second one as a dimer bound to a nucleosome. When bound to nucleosome substrate, the PRC2:EZH1 dimer undergoes a dramatic conformational change. We demonstrate that mutation of a divergent EZH1/2 loop abrogates the nucleosome-binding and methyltransferase activities of PRC2:EZH1. Finally, we show that PRC2:EZH1 dimers are more effective than monomers at promoting chromatin compaction, and the divergent EZH1/2 loop is essential for this function, thereby tying together the methyltransferase, nucleosome-binding, and chromatin-compaction activities of PRC2:EZH1. We speculate that the conformational flexibility and the ability to dimerize enable PRC2 to act on the varied chromatin substrates it encounters in the cell.
- Skirball Institute of Biomolecular Medicine, Department of Biochemistry and Molecular Pharmacology, New York University Grossman School of Medicine, New York, NY, USA.
Organizational Affiliation: