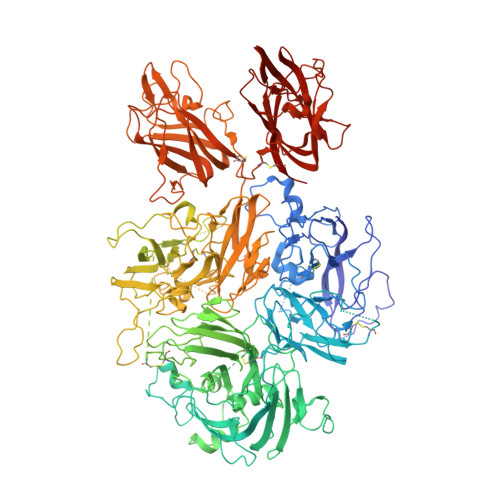
Structure of blood coagulation factor VIII in complex with an anti-C1 domain pathogenic antibody inhibitor.
Gish, J.S., Jarvis, L., Childers, K.C., Peters, S.C., Garrels, C.S., Smith, I.W., Spencer, H.T., Doering, C.B., Lollar, P., Spiegel, P.C.(2021) Blood 137: 2981-2986
- PubMed: 33529335
- DOI: https://doi.org/10.1182/blood.2020008940
- Primary Citation of Related Structures:
7K66 - PubMed Abstract:
Antibody inhibitor development in hemophilia A represents the most significant complication resulting from factor VIII (fVIII) replacement therapy. Recent studies have demonstrated that epitopes present in the C1 domain contribute to a pathogenic inhibitor response. In this study, we report the structure of a group A anti-C1 domain inhibitor, termed 2A9, in complex with a B domain-deleted, bioengineered fVIII construct (ET3i). The 2A9 epitope forms direct contacts to the C1 domain at 3 different surface loops consisting of Lys2065-Trp2070, Arg2150-Tyr2156, and Lys2110-Trp2112. Additional contacts are observed between 2A9 and the A3 domain, including the Phe1743-Tyr1748 loop and the N-linked glycosylation at Asn1810. Most of the C1 domain loops in the 2A9 epitope also represent a putative interface between fVIII and von Willebrand factor. Lastly, the C2 domain in the ET3i:2A9 complex adopts a large, novel conformational change, translocating outward from the structure of fVIII by 20 Å. This study reports the first structure of an anti-C1 domain antibody inhibitor and the first fVIII:inhibitor complex with a therapeutically active fVIII construct. Further structural understanding of fVIII immunogenicity may result in the development of more effective and safe fVIII replacement therapies.
- Department of Chemistry, Western Washington University, Bellingham, WA; and.
Organizational Affiliation: