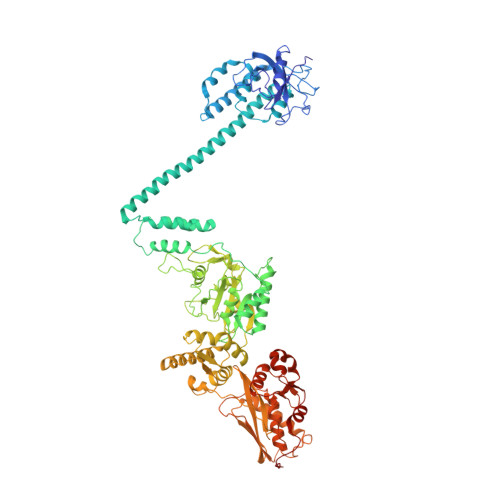
Complete three-dimensional structures of the Lon protease translocating a protein substrate.
Li, S., Hsieh, K.Y., Kuo, C.I., Lee, S.H., Pintilie, G.D., Zhang, K., Chang, C.I.(2021) Sci Adv 7: eabj7835-eabj7835
- PubMed: 34652947 Search on PubMedSearch on PubMed Central
- DOI: https://doi.org/10.1126/sciadv.abj7835
- Primary Citation Related Structures:
7FD4, 7FD5 - PubMed Abstract:
Lon is an evolutionarily conserved proteolytic machine carrying out a wide spectrum of biological activities by degrading misfolded damaged proteins and specific cellular substrates. Lon contains a large N-terminal domain and forms a hexameric core of fused adenosine triphosphatase and protease domains. Here, we report two complete structures of Lon engaging a substrate, determined by cryo–electron microscopy to 2.4-angstrom resolution. These structures show a multilayered architecture featuring a tensegrity triangle complex, uniquely constructed by six long N-terminal helices. The interlocked helix triangle is assembled on the top of the hexameric core to spread a web of six globular substrate-binding domains. It serves as a multipurpose platform that controls the access of substrates to the AAA+ ring, provides a ruler-based mechanism for substrate selection, and acts as a pulley device to facilitate unfolding of the translocated substrate. This work provides a complete framework for understanding the structural mechanisms of Lon.
- MOE Key Laboratory for Membraneless Organelles and Cellular Dynamics, Hefei National Laboratory for Physical Sciences at the Microscale and Division of Life Sciences and Medicine, University of Science and Technology of China, Hefei 230027, China.
Organizational Affiliation: