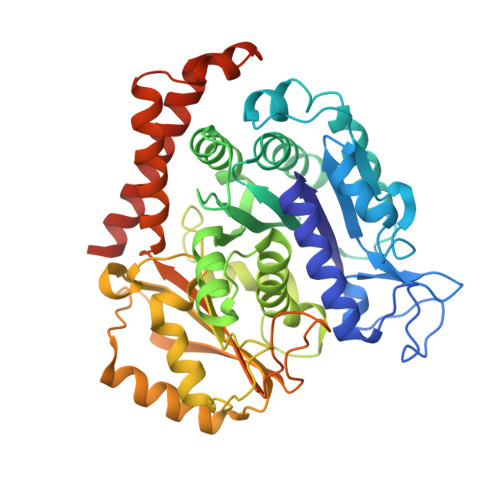
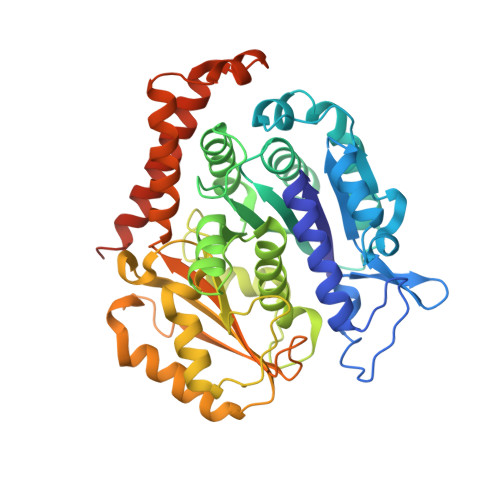
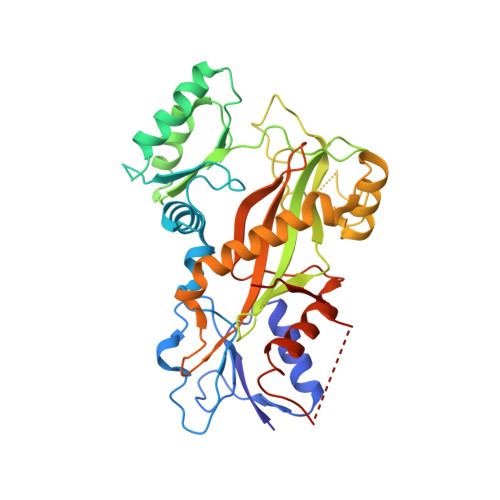
Small Molecules Promote Selective Denaturation and Degradation of Tubulin Heterodimers through a Low-Barrier Hydrogen Bond.
Yang, J., Li, Y., Qiu, Q., Wang, R., Yan, W., Yu, Y., Niu, L., Pei, H., Wei, H., Ouyang, L., Ye, H., Xu, D., Wei, Y., Chen, Q., Chen, L.(2022) J Med Chem 65: 9159-9173
- PubMed: 35762925 Search on PubMed
- DOI: https://doi.org/10.1021/acs.jmedchem.2c00379
- Primary Citation Related Structures:
7CDA, 7CE6, 7CE8, 7CEK - PubMed Abstract:
Here, we report a novel mechanism to selectively degrade target proteins. 3-(3-Phenoxybenzyl)amino-β-carboline (PAC), a tubulin inhibitor, promotes selective degradation of αβ-tubulin heterodimers. Biochemical studies have revealed that PAC specifically denatures tubulin, making it prone to aggregation that predisposes it to ubiquitinylation and then degradation. The degradation is mediated by a single hydrogen bond formed between the pyridine nitrogen of PAC and βGlu198, which is identified as a low-barrier hydrogen bond (LBHB). In contrast, another two tubulin inhibitors that only form normal hydrogen bonds with βGlu198 exhibit no degradation effect. Thus, the LBHB accounts for the degradation. We then screened for compounds capable of forming an LBHB with βGlu198 and demonstrated that BML284, a Wnt signaling activator, also promotes tubulin heterodimer degradation through the LBHB. Our study provided a unique example of LBHB function and identified a novel approach to obtain tubulin degraders.
- Laboratory of Natural and Targeted Small Molecule Drugs, State Key Laboratory of Biotherapy and Cancer Center, National Clinical Research Center for Geriatrics, West China Hospital, Sichuan University and Collaborative Innovation Center of Biotherapy, Chengdu 610041, China.
Organizational Affiliation: