Characterization and structural basis of D-cysteine desulfhydrase from Pectobacterium atrosepticum
Xu, X., Yang, L., Zhang, X., Xing, X., Zhou, J.(2022) Tetrahedron : 133174
Experimental Data Snapshot
Starting Model: experimental
View more details
wwPDB Validation 3D Report Full Report
Macromolecule Content
Entity ID: 1 | |||||
|---|---|---|---|---|---|
| Molecule | Chains | Sequence Length | Organism | Details | Image |
| D-Cysteine desulfhydrase | 371 | Pectobacterium atrosepticum SCRI1043 | Mutation(s): 0 Gene Names: ECA1531 EC: 4.4.1.15 (PDB Primary Data), 4.4.1.25 (UniProt) | 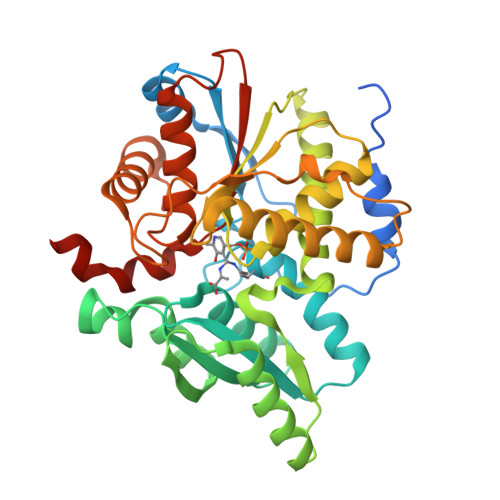 | |
UniProt | |||||
Entity Groups | |||||
| Sequence Clusters | 30% Identity50% Identity70% Identity90% Identity95% Identity100% Identity | ||||
| UniProt Group | Q6D6Z8 | ||||
Sequence AnnotationsExpand | |||||
Reference Sequence | |||||
| Ligands 2 Unique | |||||
|---|---|---|---|---|---|
| ID | Chains | Name / Formula / InChI Key | 2D Diagram | 3D Interactions | |
| EDO Download:Ideal Coordinates CCD File | D [auth A], E [auth A], I [auth A] | 1,2-ETHANEDIOL C2 H6 O2 LYCAIKOWRPUZTN-UHFFFAOYSA-N |  | ||
| FMT Download:Ideal Coordinates CCD File | C [auth A] F [auth A] G [auth A] H [auth A] J [auth A] | FORMIC ACID C H2 O2 BDAGIHXWWSANSR-UHFFFAOYSA-N |  | ||
| Modified Residues 1 Unique | |||||
|---|---|---|---|---|---|
| ID | Chains | Type | Formula | 2D Diagram | Parent |
| EXA Query on EXA | A, B | PEPTIDE LINKING | C17 H27 N4 O9 P |  | LYS |
| Length ( Å ) | Angle ( ˚ ) |
|---|---|
| a = 68.38 | α = 90 |
| b = 85.478 | β = 90 |
| c = 123.091 | γ = 90 |
| Software Name | Purpose |
|---|---|
| PHENIX | refinement |
| Aimless | data scaling |
| PDB_EXTRACT | data extraction |
| XDS | data reduction |
| PHASER | phasing |
| Funding Organization | Location | Grant Number |
|---|---|---|
| Ministry of Science and Technology (MoST, China) | China | 2019YFA09005000 |