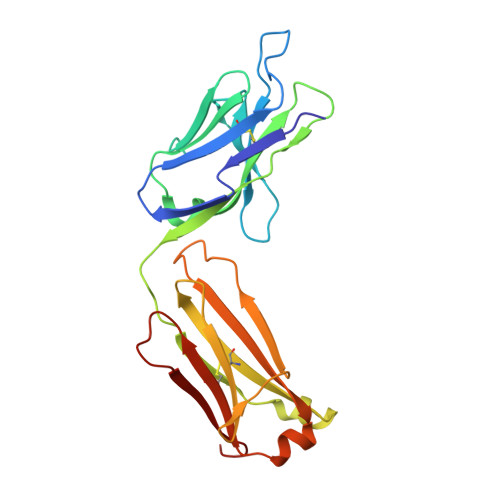
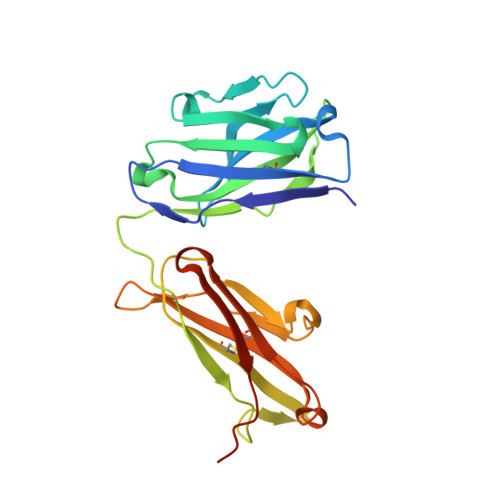
Effective rational humanization of a PASylated anti-galectin-3 Fab for the sensitive PET imaging of thyroid cancer in vivo.
Peplau, E., De Rose, F., Eichinger, A., Reder, S., Mittelhauser, M., Scafetta, G., Schwaiger, M., Weber, W.A., Bartolazzi, A., D'Alessandria, C., Skerra, A.(2021) Sci Rep 11: 7358-7358
- PubMed: 33795750 Search on PubMedSearch on PubMed Central
- DOI: https://doi.org/10.1038/s41598-021-86641-0
- Primary Citation Related Structures:
6ZVF - PubMed Abstract:
The lack of a non-invasive test for malignant thyroid nodules makes the diagnosis of thyroid cancer (TC) challenging. Human galectin-3 (hGal3) has emerged as a promising target for medical TC imaging and diagnosis because of its exclusive overexpression in malignant thyroid tissues. We previously developed a human-chimeric αhGal3 Fab fragment derived from the rat monoclonal antibody (mAb) M3/38 with optimized clearance characteristics using PASylation technology. Here, we describe the elucidation of the hGal3 epitope recognized by mAb M3/38, X-ray crystallographic analysis of its complex with the chimeric Fab and, based on the three-dimensional structure, the rational humanization of the Fab by CDR grafting. Four CDR-grafted versions were designed using structurally most closely related fully human immunoglobulin V H /V L regions of which one-employing the acceptor framework regions of the HIV-1 neutralizing human antibody m66-showed the highest antigen affinity. By introducing two additional back-mutations to the rodent donor sequence, an affinity toward hGal3 indistinguishable from the chimeric Fab was achieved (K D = 0.34 ± 0.02 nM in SPR). The PASylated humanized Fab was site-specifically labelled with the fluorescent dye Cy7 and applied for the immuno-histochemical staining of human tissue sections representative for different TCs. The same protein was conjugated with the metal chelator Dfo, followed by radiolabelling with 89 Zr(IV). The resulting protein tracer allowed the highly sensitive and specific PET/CT imaging of orthotopic tumors in mice, which was confirmed by quantitative analysis of radiotracer accumulation. Thus, the PASylated humanized αhGal3 Fab offers clinical potential for the diagnostic imaging of TC.
- Lehrstuhl für Biologische Chemie, Technische Universität München, 85354, Freising (Weihenstephan), Germany.
Organizational Affiliation: