A single-domain bispecific antibody targeting CD1d and the NKT T-cell receptor induces a potent antitumor response.
Shahine, A., Rossjohn, J.(2020) Nat Cancer
Experimental Data Snapshot
Starting Models: experimental
View more details
(2020) Nat Cancer
Macromolecule Content
Entity ID: 1 | |||||
|---|---|---|---|---|---|
| Molecule | Chains | Sequence Length | Organism | Details | Image |
| Antigen-presenting glycoprotein CD1d | 347 | Homo sapiens | Mutation(s): 0 Gene Names: CD1D | 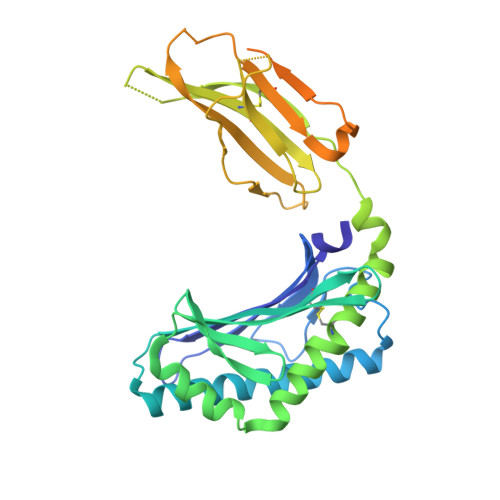 | |
UniProt & NIH Common Fund Data Resources | |||||
PHAROS: P15813 GTEx: ENSG00000158473 | |||||
Entity Groups | |||||
| Sequence Clusters | 30% Identity50% Identity70% Identity90% Identity95% Identity100% Identity | ||||
| UniProt Group | P15813 | ||||
Glycosylation | |||||
| Glycosylation Sites: 4 | Go to GlyGen: P15813-1 | ||||
Sequence AnnotationsExpand | |||||
Reference Sequence | |||||
Entity ID: 2 | |||||
|---|---|---|---|---|---|
| Molecule | Chains | Sequence Length | Organism | Details | Image |
| Beta-2-microglobulin | 100 | Homo sapiens | Mutation(s): 0 Gene Names: B2M, CDABP0092, HDCMA22P | 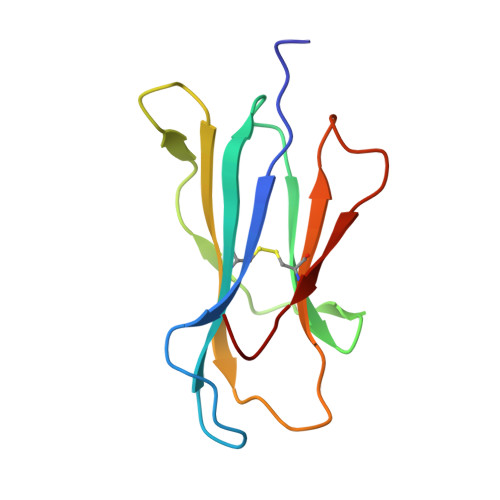 | |
UniProt & NIH Common Fund Data Resources | |||||
PHAROS: P61769 GTEx: ENSG00000166710 | |||||
Entity Groups | |||||
| Sequence Clusters | 30% Identity50% Identity70% Identity90% Identity95% Identity100% Identity | ||||
| UniProt Group | P61769 | ||||
Sequence AnnotationsExpand | |||||
Reference Sequence | |||||
Entity ID: 3 | |||||
|---|---|---|---|---|---|
| Molecule | Chains | Sequence Length | Organism | Details | Image |
| Nanobody VHH ID5 | C [auth F] | 127 | Lama glama | Mutation(s): 0 | 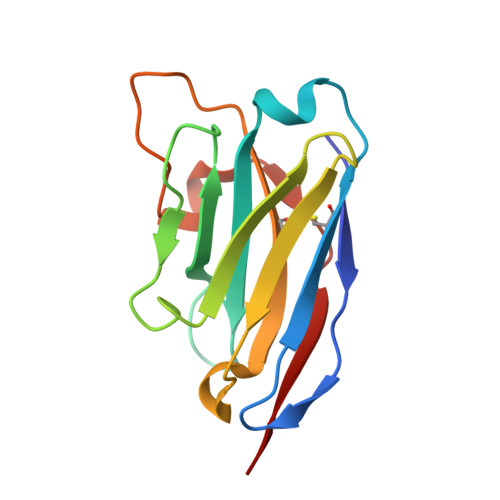 |
Entity ID: 4 | |||||
|---|---|---|---|---|---|
| Molecule | Chains | Length | 2D Diagram | Glycosylation | D Interactions |
| 2-acetamido-2-deoxy-beta-D-glucopyranose-(1-4)-2-acetamido-2-deoxy-beta-D-glucopyranose | D [auth C] | 2 |  | N-Glycosylation | |
Glycosylation Resources | |||||
GlyTouCan: G42666HT GlyCosmos: G42666HT GlyGen: G42666HT | |||||
| Ligands 4 Unique | |||||
|---|---|---|---|---|---|
| ID | Chains | Name / Formula / InChI Key | 2D Diagram | 3D Interactions | |
| AGH (Subject of Investigation/LOI) Download:Ideal Coordinates CCD File | H [auth A] | N-{(1S,2R,3S)-1-[(ALPHA-D-GALACTOPYRANOSYLOXY)METHYL]-2,3-DIHYDROXYHEPTADECYL}HEXACOSANAMIDE C50 H99 N O9 VQFKFAKEUMHBLV-BYSUZVQFSA-N |  | ||
| NAG Download:Ideal Coordinates CCD File | F [auth A], I [auth A] | 2-acetamido-2-deoxy-beta-D-glucopyranose C8 H15 N O6 OVRNDRQMDRJTHS-FMDGEEDCSA-N |  | ||
| SO4 Download:Ideal Coordinates CCD File | G [auth A], N [auth B], O [auth B], P [auth B], Q [auth B] | SULFATE ION O4 S QAOWNCQODCNURD-UHFFFAOYSA-L |  | ||
| CL Download:Ideal Coordinates CCD File | J [auth A], K [auth A], L [auth A], M [auth A], R [auth B] | CHLORIDE ION Cl VEXZGXHMUGYJMC-UHFFFAOYSA-M |  | ||
| Length ( Å ) | Angle ( ˚ ) |
|---|---|
| a = 73.27 | α = 90 |
| b = 73.27 | β = 90 |
| c = 261.077 | γ = 120 |
| Software Name | Purpose |
|---|---|
| BUSTER | refinement |
| PDB_EXTRACT | data extraction |
| XDS | data reduction |
| Aimless | data scaling |
| PHASER | phasing |