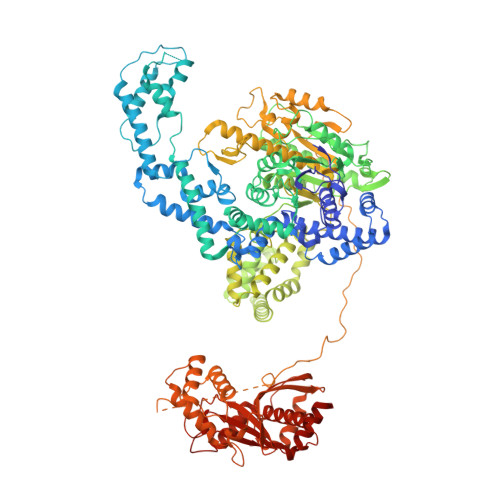
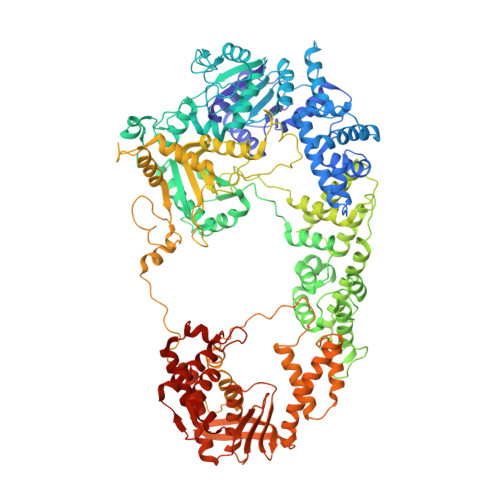
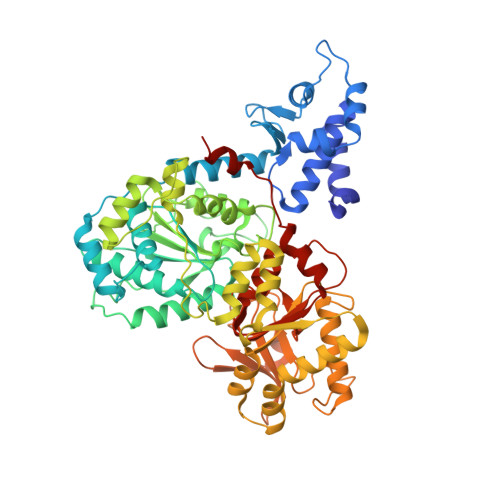
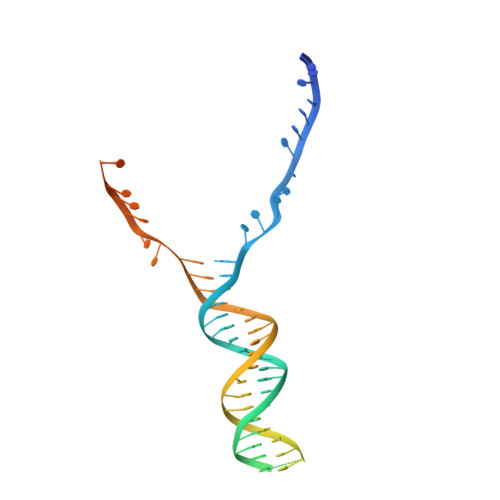
A conformational switch in response to Chi converts RecBCD from phage destruction to DNA repair.
Cheng, K., Wilkinson, M., Chaban, Y., Wigley, D.B.(2020) Nat Struct Mol Biol 27: 71-77
- PubMed: 31907455 Search on PubMedSearch on PubMed Central
- DOI: https://doi.org/10.1038/s41594-019-0355-2
- Primary Citation Related Structures:
6SJB, 6SJE, 6SJF, 6SJG, 6T2U, 6T2V - PubMed Abstract:
The RecBCD complex plays key roles in phage DNA degradation, CRISPR array acquisition (adaptation) and host DNA repair. The switch between these roles is regulated by a DNA sequence called Chi. We report cryo-EM structures of the Escherichia coli RecBCD complex bound to several different DNA forks containing a Chi sequence, including one in which Chi is recognized and others in which it is not. The Chi-recognized structure shows conformational changes in regions of the protein that contact Chi and reveals a tortuous path taken by the DNA. Sequence specificity arises from interactions with both the RecC subunit and the sequence itself. These structures provide molecular details for how Chi is recognized and insights into the changes that occur in response to Chi binding that switch RecBCD from bacteriophage destruction and CRISPR spacer acquisition to constructive host DNA repair.
- Section of Structural Biology, Department of Infectious Disease, Faculty of Medicine, Imperial College London, London, UK.
Organizational Affiliation: