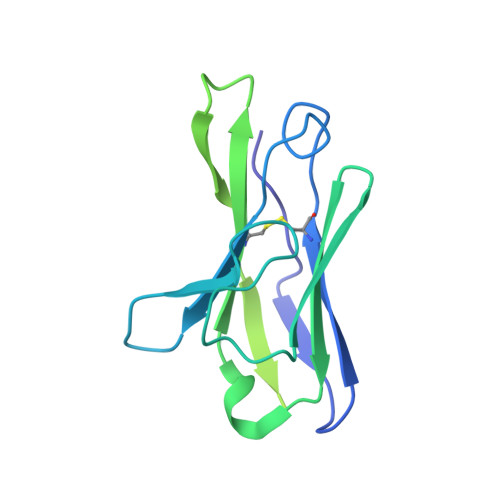
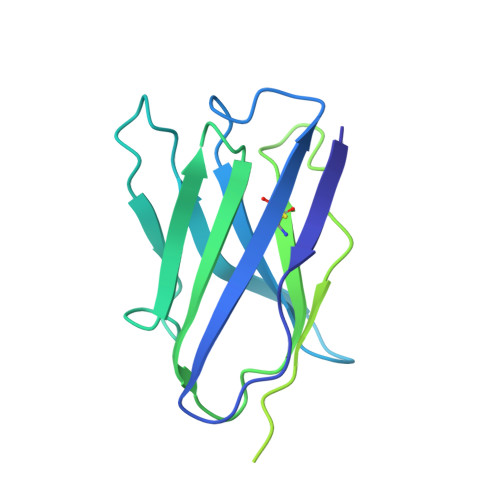
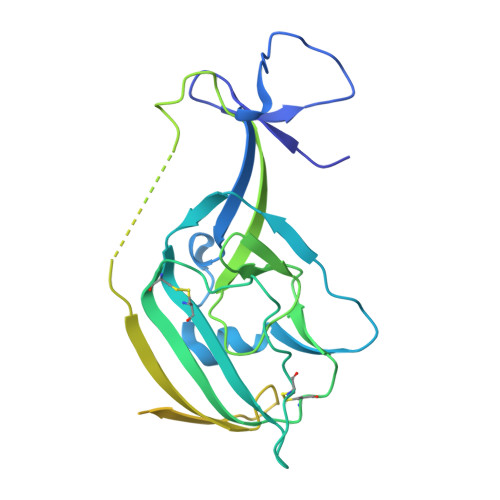
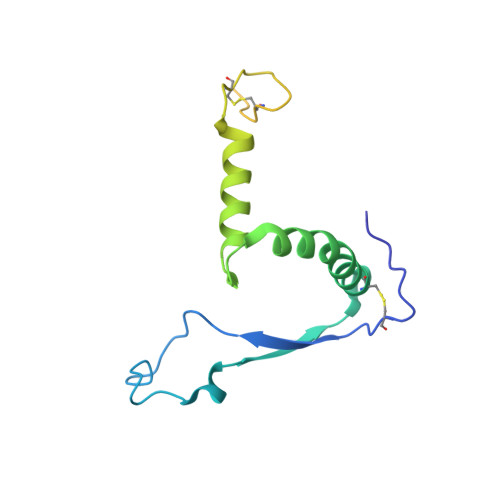
Structural Basis for a Convergent Immune Response against Ebola Virus.
Cohen-Dvashi, H., Zehner, M., Ehrhardt, S., Katz, M., Elad, N., Klein, F., Diskin, R.(2020) Cell Host Microbe 27: 418-427.e4
- PubMed: 32059794 Search on PubMed
- DOI: https://doi.org/10.1016/j.chom.2020.01.007
- Primary Citation Related Structures:
6S8D, 6S8I, 6S8J - PubMed Abstract:
Ebola virus disease is a severe health problem in Africa. Vaccines that display the Zaire ebolavirus glycoprotein spike complex are a prime component for the effort to combat it. The V H 3-15/V λ 1-40-based class of antibodies was recently discovered to be a common response in individuals who received the Ebola virus vaccines. These antibodies display attractive properties, and thus likely contribute to the efficacy of the vaccines. Here, we use cryo-EM to elucidate how three V H 3-15/V λ 1-40 antibodies from different individuals target the virus and found a convergent mechanism against a partially conserved site on the spike complex. Our study rationalizes the selection of the V H 3-15/V λ 1-40 germline genes for specifically targeting this site and highlights Ebolavirus species-specific sequence divergences that may restrict breadth of V H 3-15/V λ 1-40-based humoral response. The results from this study could help develop improved immunization schemes and further enable the design of immunogens that would be efficacious against a broader set of Ebolavirus species.
- Department of Structural Biology, Weizmann Institute of Science, Rehovot 76100, Israel.
Organizational Affiliation: