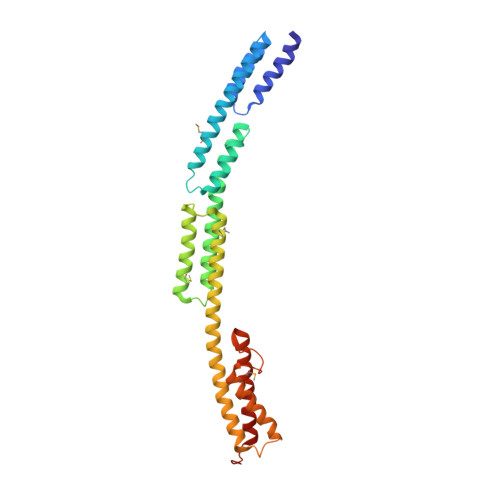
Novel structure of the N-terminal helical domain of BibA, a group B streptococcus immunogenic bacterial adhesin.
Manne, K., Chattopadhyay, D., Agarwal, V., Blom, A.M., Khare, B., Chakravarthy, S., Chang, C., Ton-That, H., Narayana, S.V.L.(2020) Acta Crystallogr D Struct Biol 76: 759-770
- PubMed: 32744258 Search on PubMedSearch on PubMed Central
- DOI: https://doi.org/10.1107/S2059798320008116
- Primary Citation Related Structures:
6POO - PubMed Abstract:
BibA, a group B streptococcus (GBS) surface protein, has been shown to protect the pathogen from phagocytic killing by sequestering a complement inhibitor: C4b-binding protein (C4BP). Here, the X-ray crystallographic structure of a GBS BibA fragment (BibA 126-398 ) and a low-resolution small-angle X-ray scattering (SAXS) structure of the full-length N-terminal domain (BibA 34-400 ) are described. The BibA 126-398 fragment crystal structure displayed a novel and predominantly helical structure. The tertiary arrangement of helices forms four antiparallel three-helix-bundle-motif repeats, with one long helix from a bundle extending into the next. Multiple mutations on recombinant BibA 34-400 delayed the degradation of the protein, and circular dichroism spectroscopy of BibA 34-400 suggested a similar secondary-structure composition to that observed in the crystallized BibA 126-398 fragment. A model was generated for the 92 N-terminal residues (BibA 34-125 ) using structural similarity prediction programs, and a BibA 34-400 model was generated by combining the coordinates of BibA 34-126 and BibA 126-398 . The X-ray structure of BibA 126-398 and the model of BibA 34-400 fitted well into the calculated SAXS envelope. One possible binding site for the BibA N-terminal domain was localized to the N-terminal CCP (complement-control protein) domains of the C4BP α-chain, as indicated by the decreased binding of BibA to a ΔCCP1 C4BP α-chain mutant. In summary, it is suggested that the GBS surface protein BibA, which consists of three antiparallel α-helical-bundle motifs, is unique and belongs to a new class of Gram-positive surface adhesins.
- Center for Biophysical Sciences and Engineering, University of Alabama at Birmingham, Birningham, AL 35294, USA.
Organizational Affiliation: