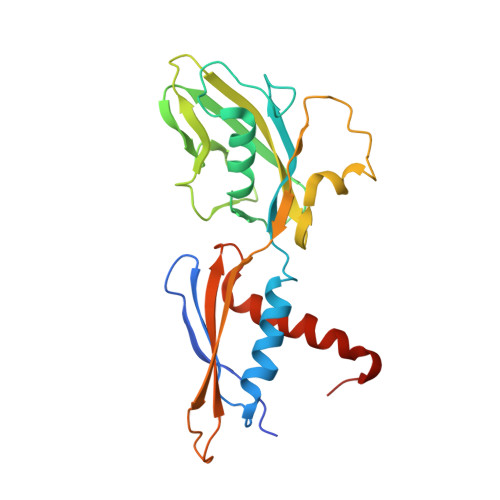
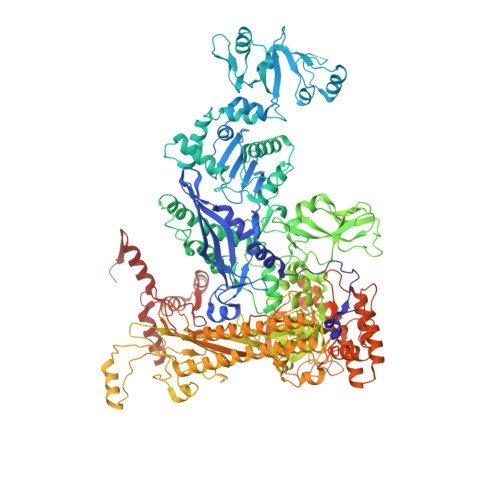
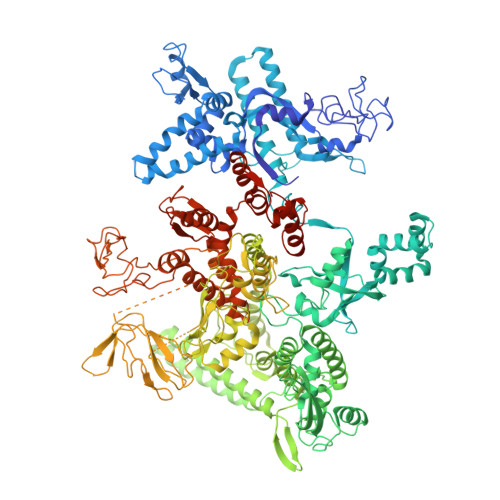
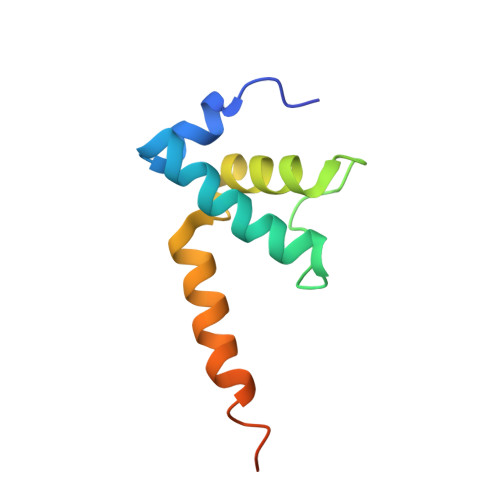
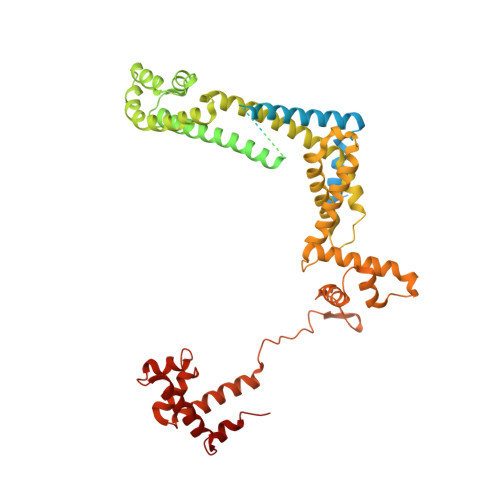
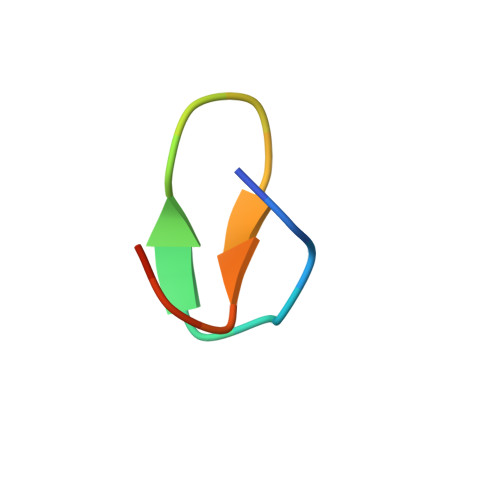
Structural mechanism of transcription inhibition by lasso peptides microcin J25 and capistruin.
Braffman, N.R., Piscotta, F.J., Hauver, J., Campbell, E.A., Link, A.J., Darst, S.A.(2019) Proc Natl Acad Sci U S A 116: 1273-1278
- PubMed: 30626643 Search on PubMedSearch on PubMed Central
- DOI: https://doi.org/10.1073/pnas.1817352116
- Primary Citation Related Structures:
6N60, 6N61, 6N62 - PubMed Abstract:
We report crystal structures of the antibacterial lasso peptides microcin J25 (MccJ25) and capistruin (Cap) bound to their natural enzymatic target, the bacterial RNA polymerase (RNAP). Both peptides bind within the RNAP secondary channel, through which NTP substrates enter the RNAP active site, and sterically block trigger-loop folding, which is essential for efficient catalysis by the RNAP. MccJ25 binds deep within the secondary channel in a manner expected to interfere with NTP substrate binding, explaining the partial competitive mechanism of inhibition with respect to NTPs found previously [Mukhopadhyay J, Sineva E, Knight J, Levy RM, Ebright RH (2004) Mol Cell 14:739-751]. The Cap binding determinant on RNAP overlaps, but is not identical to, that of MccJ25. Cap binds further from the RNAP active site and does not sterically interfere with NTP binding, and we show that Cap inhibition is partially noncompetitive with respect to NTPs. This work lays the groundwork for structure determination of other lasso peptides that target the bacterial RNAP and provides a structural foundation to guide lasso peptide antimicrobial engineering approaches.
- Laboratory of Molecular Biophysics, The Rockefeller University, New York, NY 10065.
Organizational Affiliation: