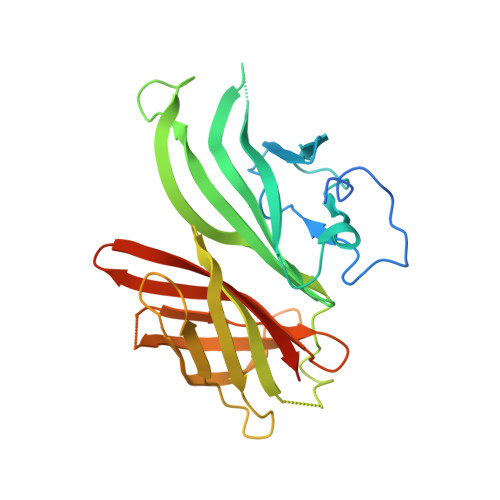
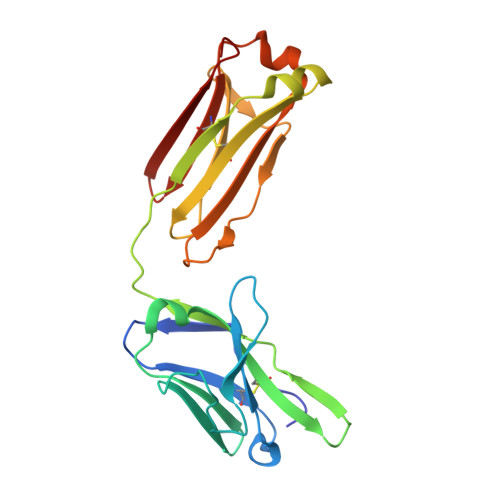
Cocrystal structure of meningococcal factor H binding protein variant 3 reveals a new crossprotective epitope recognized by human mAb 1E6.
Bianchi, F., Veggi, D., Santini, L., Buricchi, F., Bartolini, E., Lo Surdo, P., Martinelli, M., Finco, O., Masignani, V., Bottomley, M.J., Maione, D., Cozzi, R.(2019) FASEB J 33: 12099-12111
- PubMed: 31442074 Search on PubMedSearch on PubMed Central
- DOI: https://doi.org/10.1096/fj.201900374R
- Primary Citation Related Structures:
6H2Y - PubMed Abstract:
The 4 component meningococcus B vaccine (4CMenB) vaccine is the first vaccine containing recombinant proteins licensed for the prevention of invasive meningococcal disease caused by meningococcal serogroup B strains. 4CMenB contains 3 main recombinant proteins, including the Neisseria meningitidis factor H binding protein (fHbp), a lipoprotein able to bind the human factor H. To date, over 1000 aa sequences of fHbp have been identified, and they can be divided into variant groups 1, 2, and 3, which are usually not crossprotective. Nevertheless, previous characterizations of a small set ( n = 10) of mAbs generated in humans after 4CMenB immunization revealed 2 human Fabs (huFabs) (1A12, 1G3) with some crossreactivity for variants 1, 2, and 3. This unexpected result prompted us to examine a much larger set of human mAbs ( n = 110), with the aim of better understanding the extent and nature of crossreactive anti-fHbp antibodies. In this study, we report an analysis of the human antibody response to fHbp, by the characterization of 110 huFabs collected from 3 adult vaccinees during a 6-mo study. Although the 4CMenB vaccine contains fHbp variant 1, 13 huFabs were also found to be crossreactive with variants 2 and 3. The crystal structure of the crossreactive huFab 1E6 in complex with fHbp variant 3 was determined, revealing a novel, highly conserved epitope distinct from the epitopes recognized by 1A12 or 1G3. Further, functional characterization shows that human mAb 1E6 is able to elicit rabbit, but not human, complement-mediated bactericidal activity against meningococci displaying fHbp from any of the 3 different variant groups. This functional and structural information about the human antibody response upon 4CMenB immunization contributes to further unraveling the immunogenic properties of fHbp. Knowledge gained about the epitope profile recognized by the human antibody repertoire could guide future vaccine design.-Bianchi, F., Veggi, D., Santini, L., Buricchi, F., Bartolini, E., Lo Surdo, P., Martinelli, M., Finco, O., Masignani, V., Bottomley, M. J., Maione, D., Cozzi, R. Cocrystal structure of meningococcal factor H binding protein variant 3 reveals a new crossprotective epitope recognized by human mAb 1E6.
- GlaxoSmithKline, Siena, Italy.
Organizational Affiliation: