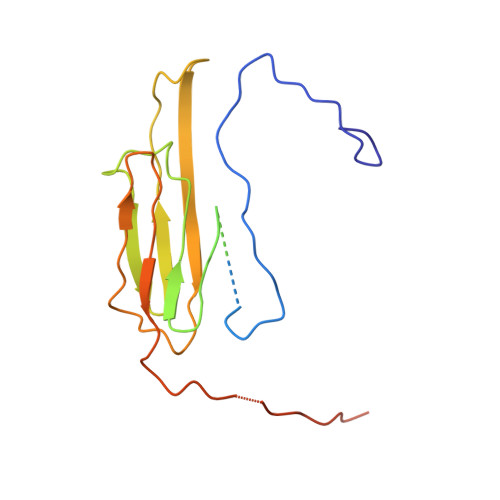
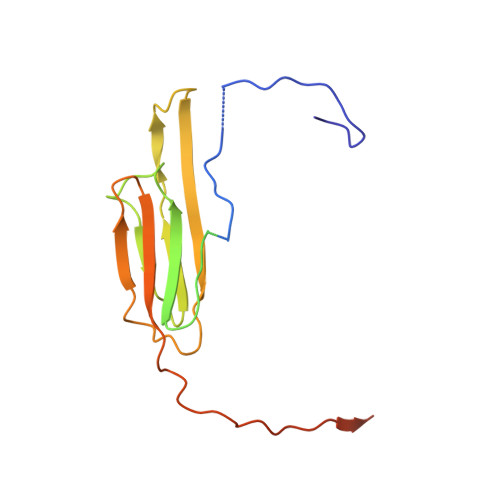
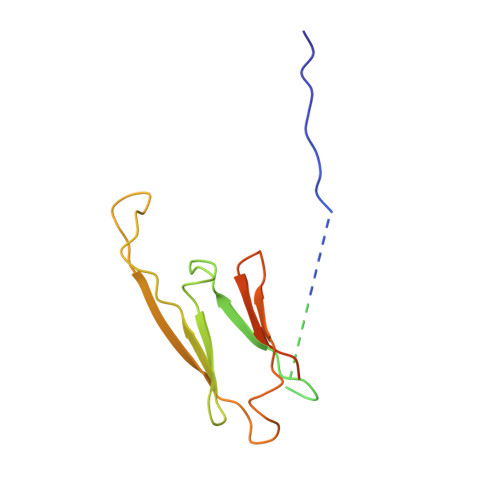
Terminal Regions Confer Plasticity to the Tetrameric Assembly of Human HspB2 and HspB3.
Clark, A.R., Vree Egberts, W., Kondrat, F.D.L., Hilton, G.R., Ray, N.J., Cole, A.R., Carver, J.A., Benesch, J.L.P., Keep, N.H., Boelens, W.C., Slingsby, C.(2018) J Mol Biology 430: 3297-3310
- PubMed: 29969581 Search on PubMedSearch on PubMed Central
- DOI: https://doi.org/10.1016/j.jmb.2018.06.047
- Primary Citation Related Structures:
6F2R - PubMed Abstract:
Heterogeneity in small heat shock proteins (sHsps) spans multiple spatiotemporal regimes-from fast fluctuations of part of the protein, to conformational variability of tertiary structure, plasticity of the interfaces, and polydispersity of the inter-converting, and co-assembling oligomers. This heterogeneity and dynamic nature of sHsps has significantly hindered their structural characterization. Atomic coordinates are particularly lacking for vertebrate sHsps, where most available structures are of extensively truncated homomers. sHsps play important roles in maintaining protein levels in the cell and therefore in organismal health and disease. HspB2 and HspB3 are vertebrate sHsps that are found co-assembled in neuromuscular cells, and variants thereof are associated with disease. Here, we present the structure of human HspB2/B3, which crystallized as a hetero-tetramer in a 3:1 ratio. In the HspB2/B3 tetramer, the four α-crystallin domains (ACDs) assemble into a flattened tetrahedron which is pierced by two non-intersecting approximate dyads. Assembly is mediated by flexible "nuts and bolts" involving IXI/V motifs from terminal regions filling ACD pockets. Parts of the N-terminal region bind in an unfolded conformation into the anti-parallel shared ACD dimer grooves. Tracts of the terminal regions are not resolved, most likely due to their disorder in the crystal lattice. This first structure of a full-length human sHsp heteromer reveals the heterogeneous interactions of the terminal regions and suggests a plasticity that is important for the cytoprotective functions of sHsps.
- Department of Biological Sciences, Crystallography, Institute of Structural & Molecular Biology, Birkbeck College, Malet Street, London, WC1E 7HX, UK.
Organizational Affiliation: