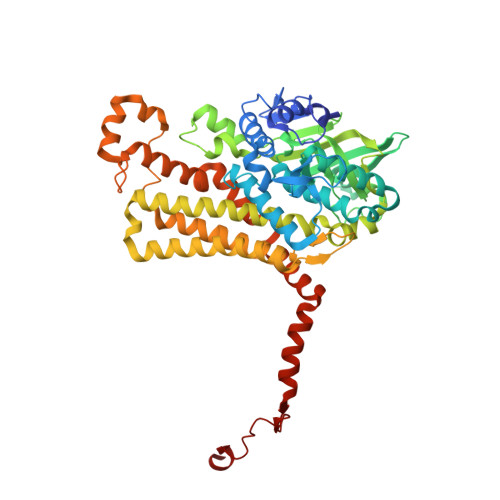
Structural Insights into Catalytic Versatility of the Flavin-dependent Hydroxylase (HpaB) from Escherichia coli.
Shen, X., Zhou, D., Lin, Y., Wang, J., Gao, S., Kandavelu, P., Zhang, H., Zhang, R., Wang, B.C., Rose, J., Yuan, Q., Yan, Y.(2019) Sci Rep 9: 7087-7087
- PubMed: 31068633 Search on PubMedSearch on PubMed Central
- DOI: https://doi.org/10.1038/s41598-019-43577-w
- Primary Citation Related Structures:
6B1B, 6EB0 - PubMed Abstract:
4-Hydroxyphenylacetate 3-hydroxylase (EcHpaB) from Escherichia coli is capable of efficient ortho-hydroxylation of a wide range of phenolic compounds and demonstrates great potential for broad chemoenzymatic applications. To understand the structural and mechanistic basis of its catalytic versatility, we elucidated the crystal structure of EcHpaB by X-ray crystallography, which revealed a unique loop structure covering the active site. We further performed mutagenesis studies of this loop to probe its role in substrate specificity and catalytic activity. Our results not only showed the loop has great plasticity and strong tolerance towards extensive mutagenesis, but also suggested a flexible loop that enables the entrance and stable binding of substrates into the active site is the key factor to the enzyme catalytic versatility. These findings lay the groundwork for editing the loop sequence and structure for generation of EcHpaB mutants with improved performance for broader laboratory and industrial use.
- Beijing Advanced Innovation Center for Soft Matter Science and Engineering, Beijing University of Chemical Technology, Beijing, 100029, China.
Organizational Affiliation: