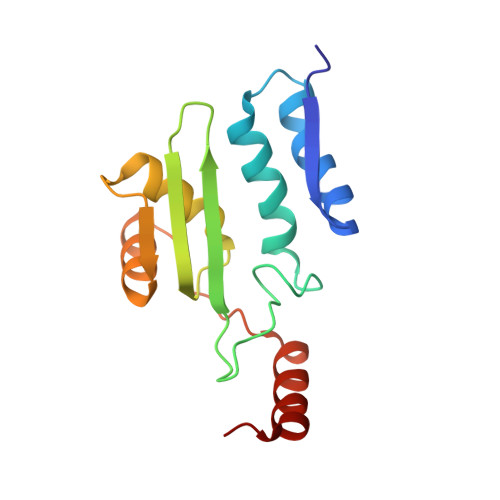
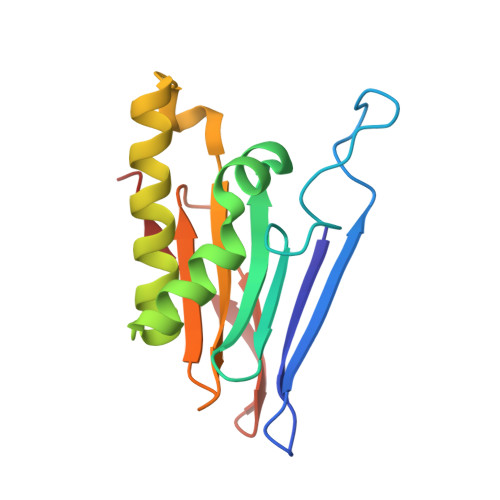
Biochemical and structural insights into an allelic variant causing the lysosomal storage disorder - aspartylglucosaminuria.
Pande, S., Bizilj, W., Guo, H.C.(2018) FEBS Lett 592: 2550-2561
- PubMed: 29993127 Search on PubMedSearch on PubMed Central
- DOI: https://doi.org/10.1002/1873-3468.13190
- Primary Citation Related Structures:
6DEY - PubMed Abstract:
Aspartylglucosaminuria (AGU) is a lysosomal storage disorder caused by defects of the hydrolase glycosylasparaginase (GA). Previously, we showed that a Canadian AGU mutation disrupts an obligatory intramolecular autoprocessing with the enzyme trapped as an inactive precursor. Here, we report biochemical and structural characterizations of a model enzyme corresponding to a Finnish AGU allele, the T234I variant. Unlike the Canadian counterpart, the Finnish variant is capable of a slow autoprocessing to generate detectible hydrolyzation activity of the natural substrate of GA. We have determined a 1.6 Å-resolution structure of the Finnish AGU model and built an enzyme-substrate complex to provide a structural basis for analyzing the negative effects of the point mutation on K M and k cat of the mature enzyme. Glycosylasparaginase or aspartylglucosaminidase, EC3.5.1.26.
- Department of Biological Sciences, University of Massachusetts Lowell, MA, USA.
Organizational Affiliation: