Non-catalytic-Region Mutations Conferring Transition of Class A beta-Lactamases Into ESBLs.
Cao, T.-P., Yi, H., Dhanasingh, I., Ghosh, S., Choi, J.M., Lee, K.H., Ryu, S., Kim, H.S., Lee, S.H.(2020) Front Mol Biosci
Experimental Data Snapshot
Starting Model: experimental
View more details
(2020) Front Mol Biosci
Macromolecule Content
Entity ID: 1 | |||||
|---|---|---|---|---|---|
| Molecule | Chains | Sequence Length | Organism | Details | Image |
| Beta-lactamase | 268 | Burkholderia thailandensis | Mutation(s): 1 Gene Names: A8H35_31635 EC: 3.5.2.6 | 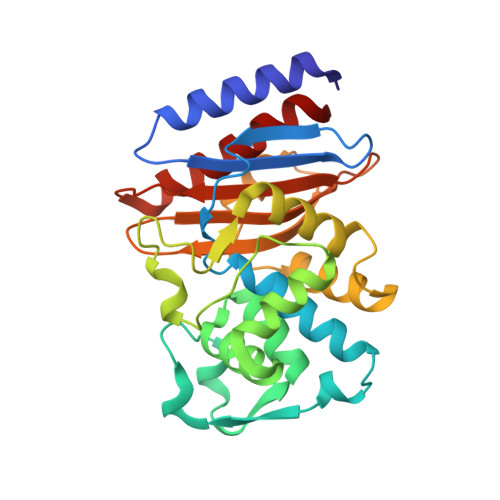 | |
UniProt | |||||
Entity Groups | |||||
| Sequence Clusters | 30% Identity50% Identity70% Identity90% Identity95% Identity100% Identity | ||||
| UniProt Group | Q2T5A3 | ||||
Sequence AnnotationsExpand | |||||
Reference Sequence | |||||
| Ligands 2 Unique | |||||
|---|---|---|---|---|---|
| ID | Chains | Name / Formula / InChI Key | 2D Diagram | 3D Interactions | |
| CB4 Download:Ideal Coordinates CCD File | B [auth A] | PINACOL[[2-AMINO-ALPHA-(1-CARBOXY-1-METHYLETHOXYIMINO)-4-THIAZOLEACETYL]AMINO]METHANEBORONATE C10 H15 B N4 O6 S ZECCQELUYUPTSB-UUASQNMZSA-N |  | ||
| GOL Download:Ideal Coordinates CCD File | C [auth A] | GLYCEROL C3 H8 O3 PEDCQBHIVMGVHV-UHFFFAOYSA-N |  | ||
| Length ( Å ) | Angle ( ˚ ) |
|---|---|
| a = 38.66 | α = 90 |
| b = 53.238 | β = 90 |
| c = 122.981 | γ = 90 |
| Software Name | Purpose |
|---|---|
| PHENIX | refinement |
| HKL-2000 | data reduction |
| HKL-2000 | data scaling |
| MOLREP | phasing |
| Funding Organization | Location | Grant Number |
|---|---|---|
| National Research Foundation (Korea) | Korea, Republic Of | NRF-2016R1D1A1B03932717 |