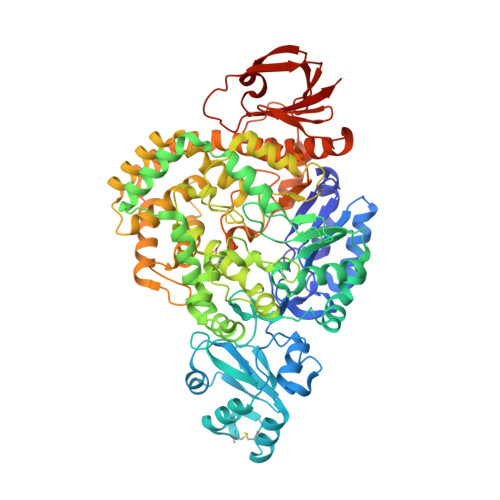
Crystal structure of glycosyltrehalose synthase from Sulfolobus shibatae DSM5389
Okazaki, N., Blaber, M., Kuroki, R., Tamada, T.(2018) Acta Crystallogr F Struct Biol Commun 74: 741-746
- PubMed: 30387780
- DOI: https://doi.org/10.1107/S2053230X1801453X
- Primary Citation of Related Structures:
5ZCR - PubMed Abstract:
Glycosyltrehalose synthase (GTSase) converts the glucosidic bond between the last two glucose residues of amylose from an α-1,4 bond to an α-1,1 bond, generating a nonreducing glycosyl trehaloside, in the first step of the biosynthesis of trehalose. To better understand the structural basis of the catalytic mechanism, the crystal structure of GTSase from the hyperthermophilic archaeon Sulfolobus shibatae DSM5389 (5389-GTSase) has been determined to 2.4 Å resolution by X-ray crystallography. The structure of 5389-GTSase can be divided into five domains. The central domain contains the (β/α) 8 -barrel fold that is conserved as the catalytic domain in the α-amylase family. Three invariant catalytic carboxylic amino acids in the α-amylase family are also found in GTSase at positions Asp241, Glu269 and Asp460 in the catalytic domain. The shape of the catalytic cavity and the pocket size at the bottom of the cavity correspond to the intramolecular transglycosylation mechanism proposed from previous enzymatic studies.
- Neutron Science and Technology Center, Comprehensive Research Organization for Science and Society, 162-1 Shirakata, Tokai, Ibaraki 319-1106, Japan.
Organizational Affiliation: