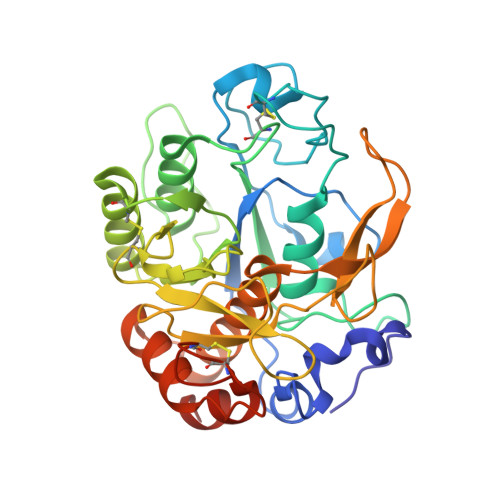
Crystal structure of a cold-active protease (Pro21717) from the psychrophilic bacterium, Pseudoalteromonas arctica PAMC 21717, at 1.4 angstrom resolution: Structural adaptations to cold and functional analysis of a laundry detergent enzyme
Park, H.J., Lee, C.W., Kim, D., Do, H., Han, S.J., Kim, J.E., Koo, B.H., Lee, J.H., Yim, J.H.(2018) PLoS One 13: e0191740-e0191740
- PubMed: 29466378 Search on PubMedSearch on PubMed Central
- DOI: https://doi.org/10.1371/journal.pone.0191740
- Primary Citation Related Structures:
5YL7 - PubMed Abstract:
Enzymes isolated from organisms found in cold habitats generally exhibit higher catalytic activity at low temperatures than their mesophilic homologs and are therefore known as cold-active enzymes. Cold-active proteases are very useful in a variety of biotechnological applications, particularly as active ingredients in laundry and dishwashing detergents, where they provide strong protein-degrading activity in cold water. We identified a cold-active protease (Pro21717) from a psychrophilic bacterium, Pseudoalteromonas arctica PAMC 21717, and determined the crystal structure of its catalytic domain (CD) at a resolution of 1.4 Å. The Pro21717-CD structure shows a conserved subtilisin-like fold with a typical catalytic triad (Asp185, His244, and Ser425) and contains four calcium ions and three disulfide bonds. Interestingly, we observed an unexpected electron density at the substrate-binding site from a co-purified peptide. Although the sequence of this peptide is unknown, analysis of the peptide-complexed structure nonetheless provides some indication of the substrate recognition and binding mode of Pro21717. Moreover, various parameters, including a wide substrate pocket size, an abundant active-site loop content, and a flexible structure provide potential explanations for the cold-adapted properties of Pro21717. In conclusion, this is first structural characterization of a cold-adapted subtilisin-like protease, and these findings provide a structural and functional basis for industrial applications of Pro21717 as a cold-active laundry or dishwashing detergent enzyme.
- Division of Polar Life Sciences, Korea Polar Research Institute, Incheon, Republic of Korea.
Organizational Affiliation: