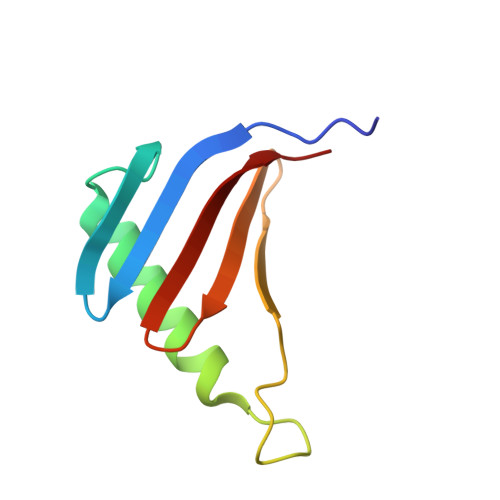
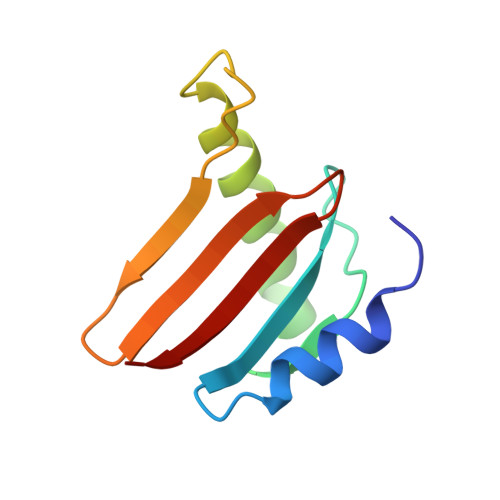
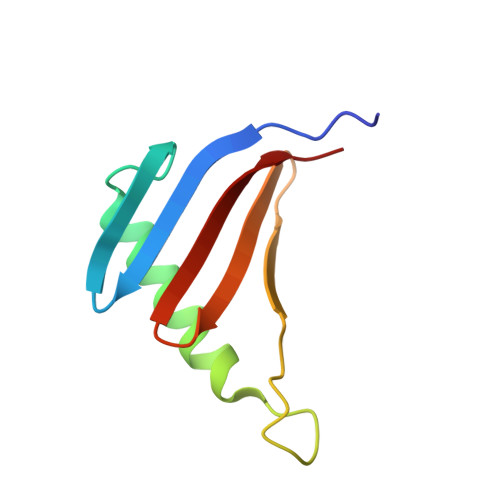
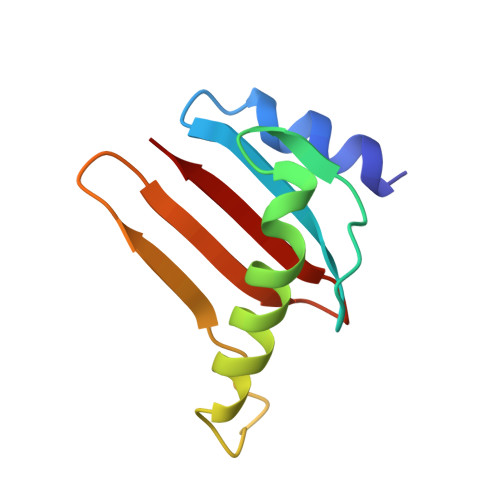
Structural insight into the Ragulator complex which anchors mTORC1 to the lysosomal membrane
Mu, Z., Wang, L., Deng, W., Wang, J., Wu, G.(2017) Cell Discov 3: 17049-17049
- PubMed: 29285400 Search on PubMedSearch on PubMed Central
- DOI: https://doi.org/10.1038/celldisc.2017.49
- Primary Citation Related Structures:
5YK3, 5YK5 - PubMed Abstract:
The mechanistic target of rapamycin (mTOR) signal-transduction pathway plays a key role in regulating many aspects of metabolic processes. The central player of the mTOR signaling pathway, mTOR complex 1 (mTORC1), is recruited by the pentameric Ragulator complex and the heterodimeric Rag GTPase complex to the lysosomal membrane and thereafter activated. Here, we determined the crystal structure of the human Ragulator complex, which shows that Lamtor1 possesses a belt-like shape and wraps the other four subunits around. Extensive hydrophobic interactions occur between Lamtor1 and the Lamtor2-Lamtor3, Lamtor4-Lamtor5 roadblock domain protein pairs, while there is no substantial contact between Lamtor2-Lamtor3 and Lamtor4-Lamtor5 subcomplexes. Interestingly, an α-helix from Lamtor1 occupies each of the positions on Lamtor4 and Lamtor5 equivalent to the α3-helices of Lamtor2 and Lamtor3, thus stabilizing Lamtor4 and Lamtor5. Structural comparison between Ragulator and the yeast Ego1-Ego2-Ego3 ternary complex (Ego-TC) reveals that Ego-TC only corresponds to half of the Ragulator complex. Coupling with the fact that in the Ego-TC structure, Ego2 and Ego3 are lone roadblock domain proteins without another roadblock domain protein pairing with them, we suggest that additional components of the yeast Ego complex might exist.
- State Key Laboratory of Microbial Metabolism, School of Life Sciences and Biotechnology, Shanghai Jiao Tong University, Shanghai, China.
Organizational Affiliation: