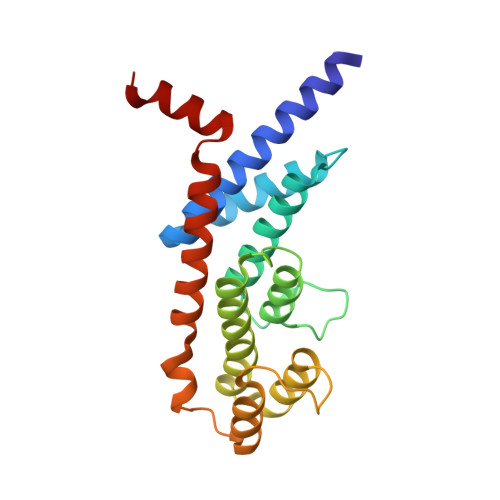
Telomeric TERB1-TRF1 interaction is crucial for male meiosis.
Long, J., Huang, C., Chen, Y., Zhang, Y., Shi, S., Wu, L., Liu, Y., Liu, C., Wu, J., Lei, M.(2017) Nat Struct Mol Biol 24: 1073-1080
- PubMed: 29083416 Search on PubMedSearch on PubMed Central
- DOI: https://doi.org/10.1038/nsmb.3496
- Primary Citation Related Structures:
5XUP - PubMed Abstract:
During meiotic prophase, the meiosis-specific telomere-binding protein TERB1 regulates chromosome movement required for homologous pairing and recombination by interacting with the telomeric shelterin subunit TRF1. Here, we report the crystal structure of the TRF1-binding motif of human TERB1 in complex with the TRFH domain of TRF1. Notably, specific disruption of the TERB1-TRF1 interaction by a point mutation in the mouse Terb1 gene results in infertility only in males. We find that this mutation causes an arrest in the zygotene-early pachytene stage and mild telomere abnormalities of autosomes but unpaired X and Y chromosomes in pachytene, leading to massive spermatocyte apoptosis. We propose that the loss of telomere structure mediated by the TERB1-TRF1 interaction significantly affects homologous pairing of the telomere-adjacent pseudoautosomal region (PAR) of the X and Y chromosomes in mouse spermatocytes. Our findings uncover a specific mechanism of telomeres that surmounts the unique challenges of mammalian X-Y pairing in meiosis.
- National Center for Protein Science Shanghai, State Key Laboratory of Molecular Biology, Institute of Biochemistry and Cell Biology, Shanghai Institutes for Biological Sciences, Chinese Academy of Sciences, Shanghai, China.
Organizational Affiliation: