The Pyruvate and alpha-Ketoglutarate Dehydrogenase Complexes of Pseudomonas aeruginosa Catalyze Pyocyanin and Phenazine-1-carboxylic Acid Reduction via the Subunit Dihydrolipoamide Dehydrogenase.
Glasser, N.R., Wang, B.X., Hoy, J.A., Newman, D.K.(2017) J Biological Chem 292: 5593-5607
- PubMed: 28174304
- DOI: https://doi.org/10.1074/jbc.M116.772848
- Primary Citation Related Structures:
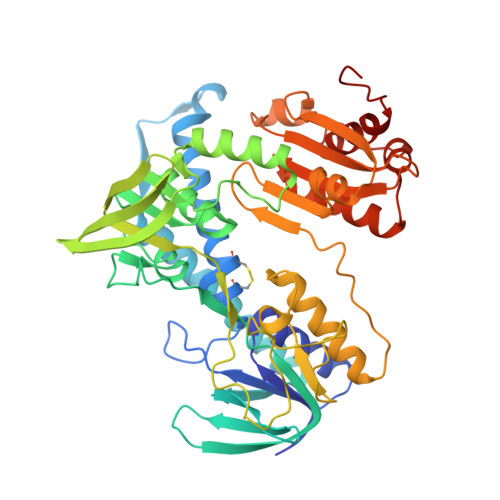
5U8U, 5U8V, 5U8W - PubMed Abstract:
Phenazines are a class of redox-active molecules produced by diverse bacteria and archaea. Many of the biological functions of phenazines, such as mediating signaling, iron acquisition, and redox homeostasis, derive from their redox activity. Although prior studies have focused on extracellular phenazine oxidation by oxygen and iron, here we report a search for reductants and catalysts of intracellular phenazine reduction in Pseudomonas aeruginosa Enzymatic assays in cell-free lysate, together with crude fractionation and chemical inhibition, indicate that P. aeruginosa contains multiple enzymes that catalyze the reduction of the endogenous phenazines pyocyanin and phenazine-1-carboxylic acid in both cytosolic and membrane fractions. We used chemical inhibitors to target general enzyme classes and found that an inhibitor of flavoproteins and heme-containing proteins, diphenyleneiodonium, effectively inhibited phenazine reduction in vitro , suggesting that most phenazine reduction derives from these enzymes. Using natively purified proteins, we demonstrate that the pyruvate and α-ketoglutarate dehydrogenase complexes directly catalyze phenazine reduction with pyruvate or α-ketoglutarate as electron donors. Both complexes transfer electrons to phenazines through the common subunit dihydrolipoamide dehydrogenase, a flavoprotein encoded by the gene lpdG Although we were unable to co-crystallize LpdG with an endogenous phenazine, we report its X-ray crystal structure in the apo-form (refined to 1.35 Å), bound to NAD + (1.45 Å), and bound to NADH (1.79 Å). In contrast to the notion that phenazines support intracellular redox homeostasis by oxidizing NADH, our work suggests that phenazines may substitute for NAD + in LpdG and other enzymes, achieving the same end by a different mechanism.
- From the Divisions of Biology and Biological Engineering and.
Organizational Affiliation: