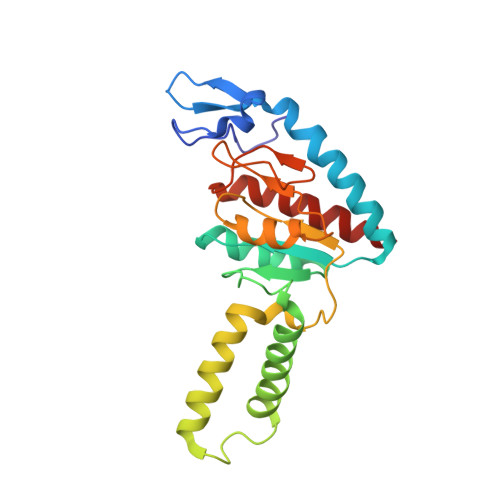
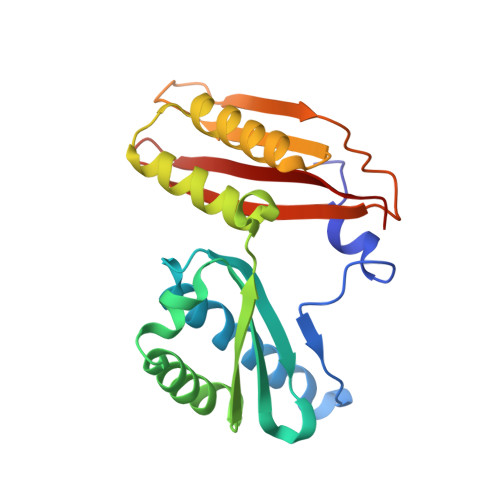
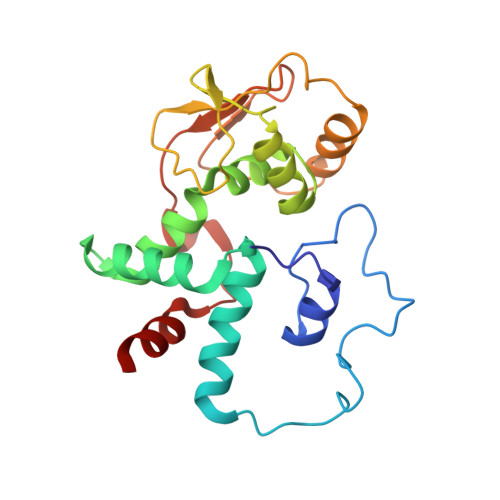
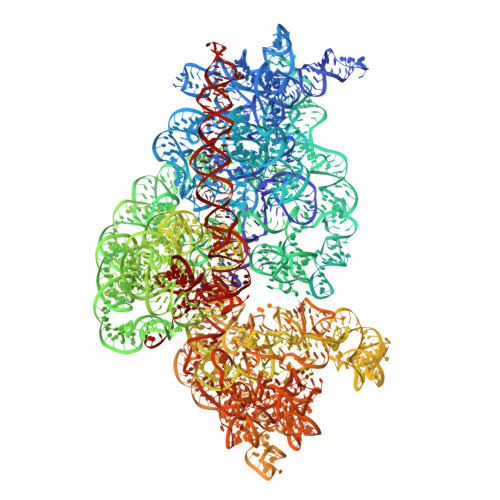
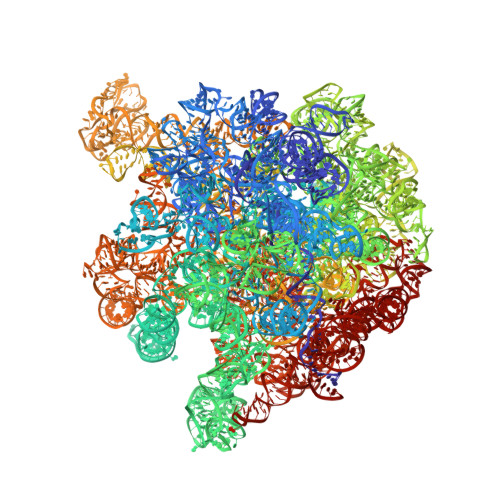
Ribosome rearrangements at the onset of translational bypassing.
Agirrezabala, X., Samatova, E., Klimova, M., Zamora, M., Gil-Carton, D., Rodnina, M.V., Valle, M.(2017) Sci Adv 3: e1700147-e1700147
- PubMed: 28630923 Search on PubMedSearch on PubMed Central
- DOI: https://doi.org/10.1126/sciadv.1700147
- Primary Citation Related Structures:
5NP6 - PubMed Abstract:
Bypassing is a recoding event that leads to the translation of two distal open reading frames into a single polypeptide chain. We present the structure of a translating ribosome stalled at the bypassing take-off site of gene 60 of bacteriophage T4. The nascent peptide in the exit tunnel anchors the P-site peptidyl-tRNA Gly to the ribosome and locks an inactive conformation of the peptidyl transferase center (PTC). The mRNA forms a short dynamic hairpin in the decoding site. The ribosomal subunits adopt a rolling conformation in which the rotation of the small subunit around its long axis causes the opening of the A-site region. Together, PTC conformation and mRNA structure safeguard against premature termination and read-through of the stop codon and reconfigure the ribosome to a state poised for take-off and sliding along the noncoding mRNA gap.
- Structural Biology Unit, CIC bioGUNE, 48160 Derio, Spain.
Organizational Affiliation: