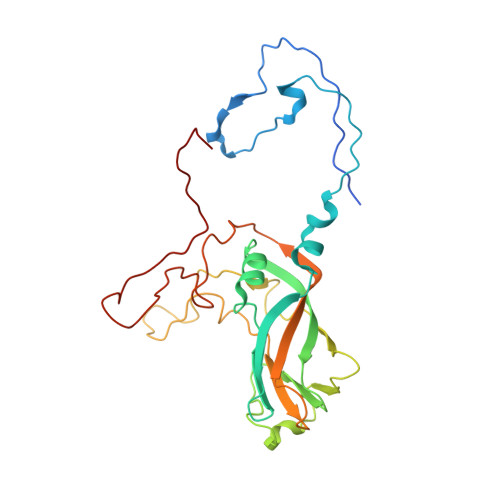
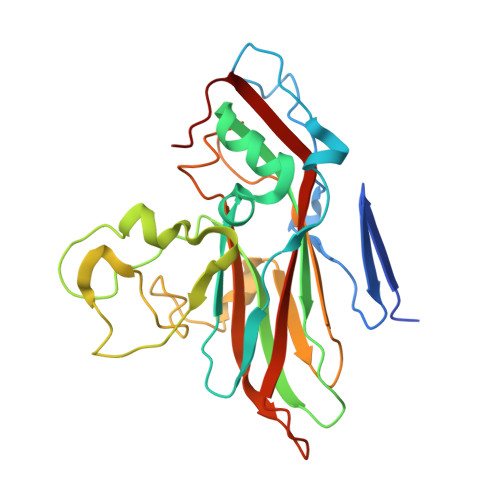
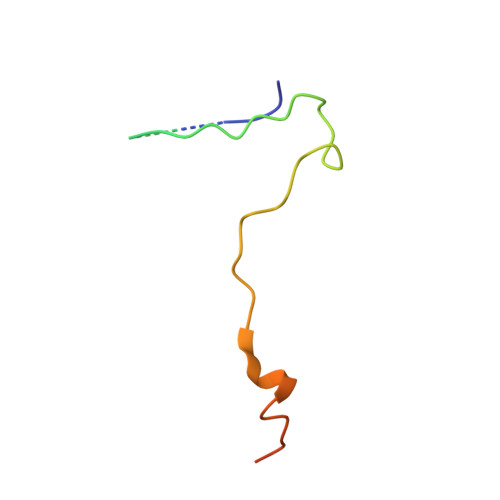
Atomic structure of a rhinovirus C, a virus species linked to severe childhood asthma.
Liu, Y., Hill, M.G., Klose, T., Chen, Z., Watters, K., Bochkov, Y.A., Jiang, W., Palmenberg, A.C., Rossmann, M.G.(2016) Proc Natl Acad Sci U S A 113: 8997-9002
- PubMed: 27511920 Search on PubMedSearch on PubMed Central
- DOI: https://doi.org/10.1073/pnas.1606595113
- Primary Citation Related Structures:
5JZG, 5K0U - PubMed Abstract:
Isolates of rhinovirus C (RV-C), a recently identified Enterovirus (EV) species, are the causative agents of severe respiratory infections among children and are linked to childhood asthma exacerbations. The RV-C have been refractory to structure determination because they are difficult to propagate in vitro. Here, we report the cryo-EM atomic structures of the full virion and native empty particle (NEP) of RV-C15a. The virus has 60 "fingers" on the virus outer surface that probably function as dominant immunogens. Because the NEPs also display these fingers, they may have utility as vaccine candidates. A sequence-conserved surface depression adjacent to each finger forms a likely binding site for the sialic acid on its receptor. The RV-C, unlike other EVs, are resistant to capsid-binding antiviral compounds because the hydrophobic pocket in VP1 is filled with multiple bulky residues. These results define potential molecular determinants for designing antiviral therapeutics and vaccines.
- Department of Biological Sciences, Purdue University, West Lafayette, IN 47907;
Organizational Affiliation: