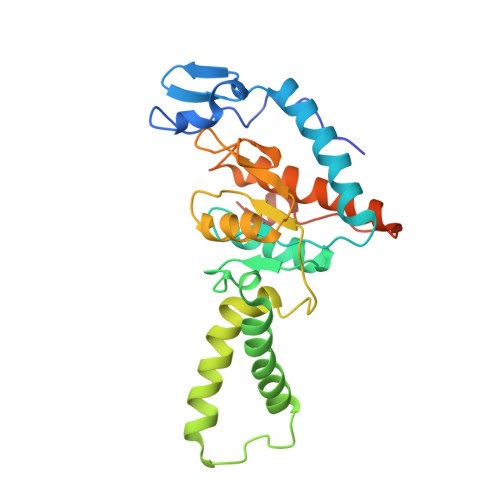
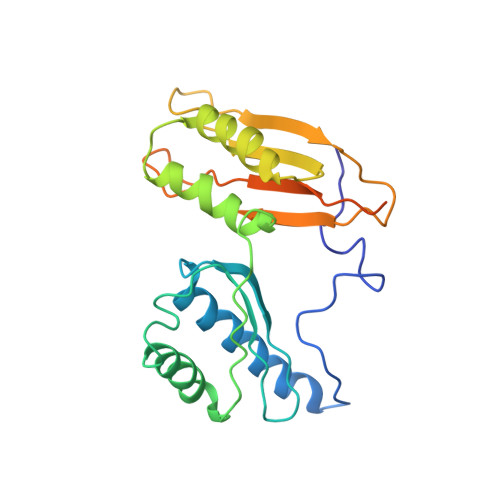
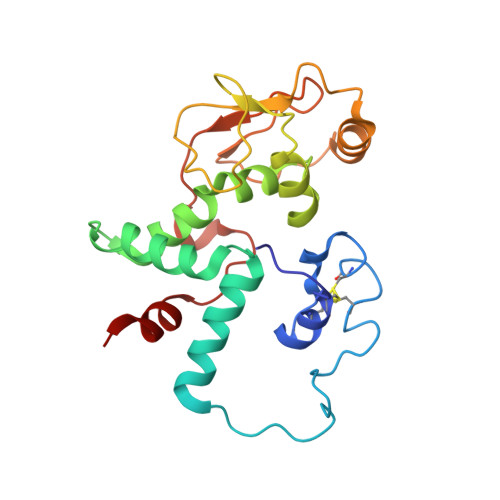
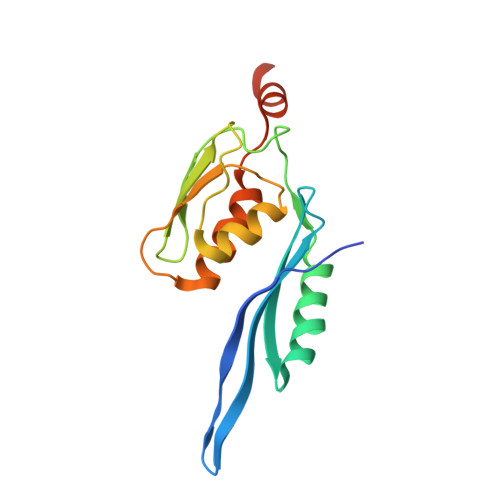
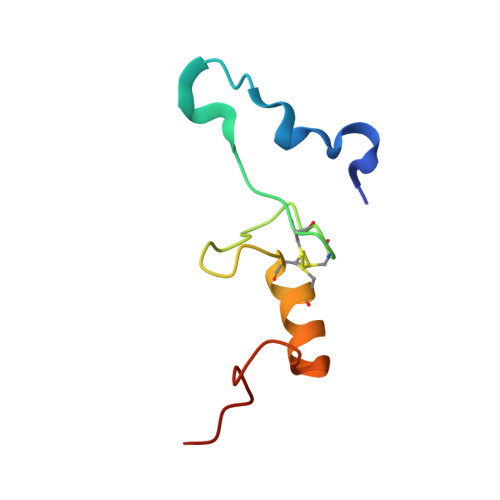
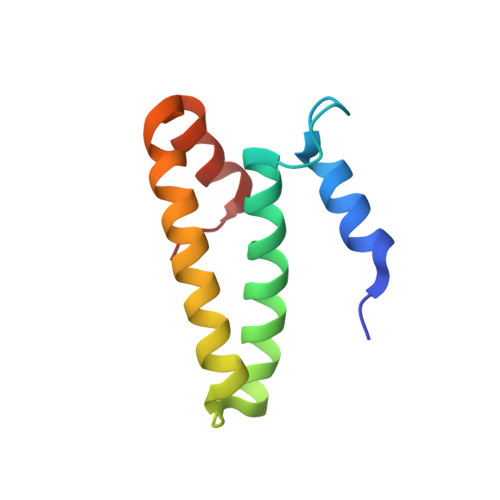
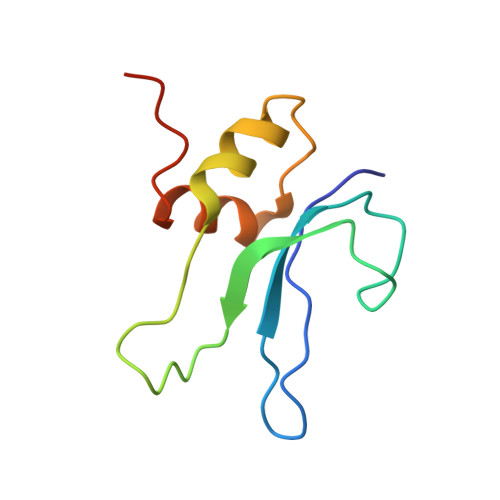
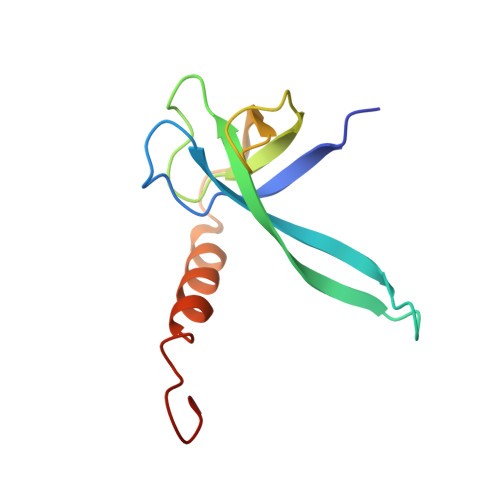
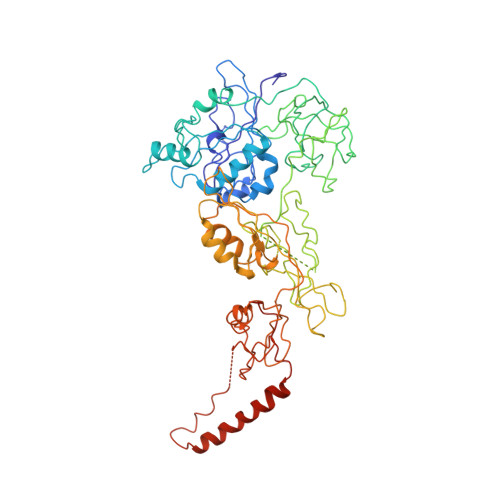
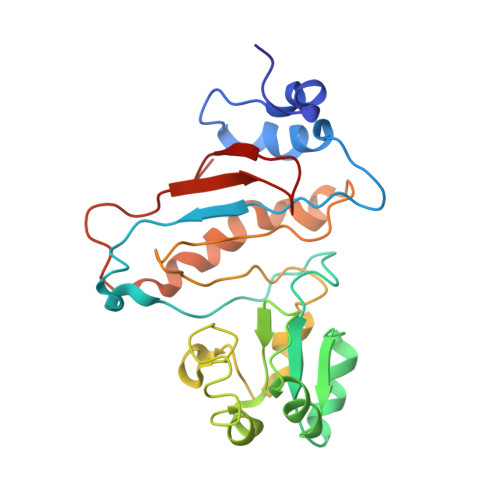
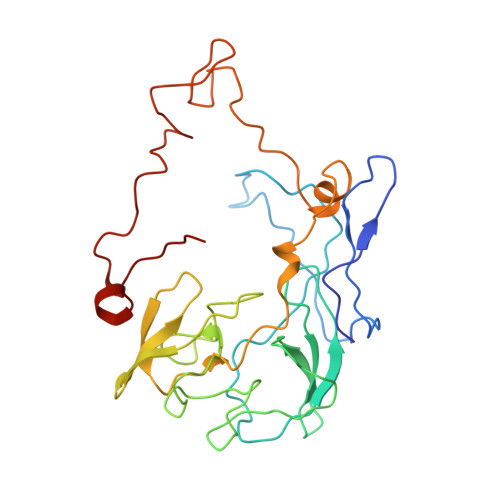
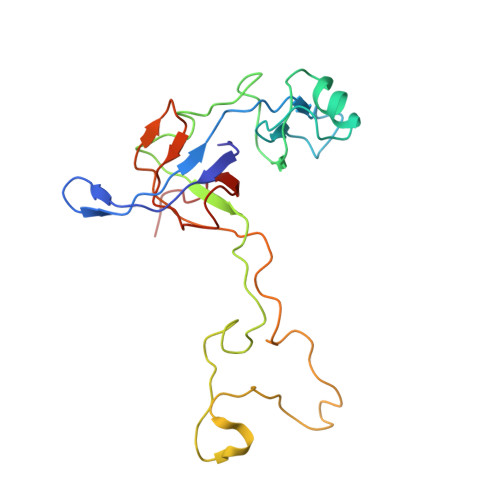
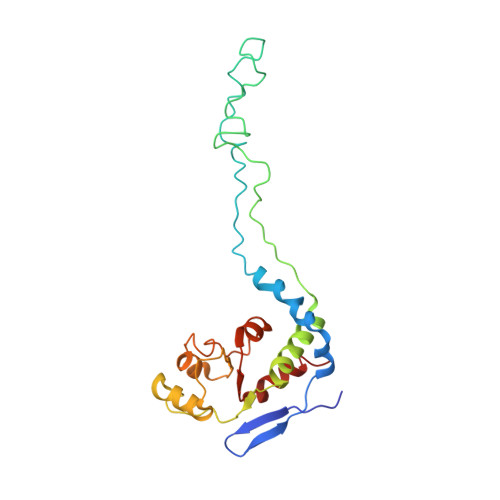
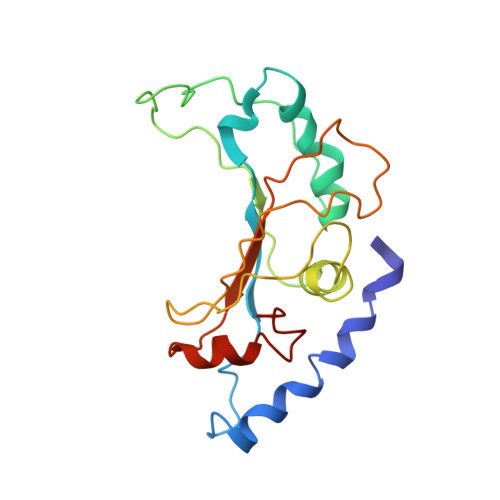
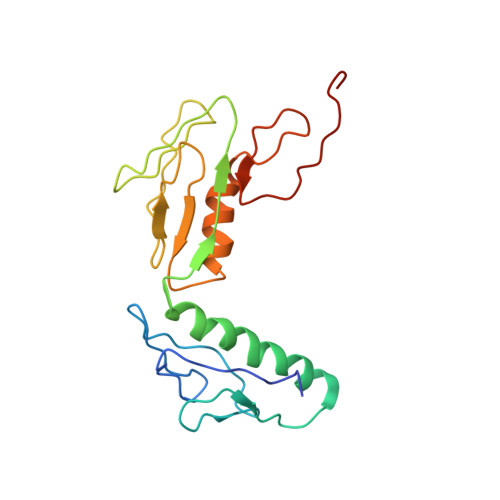
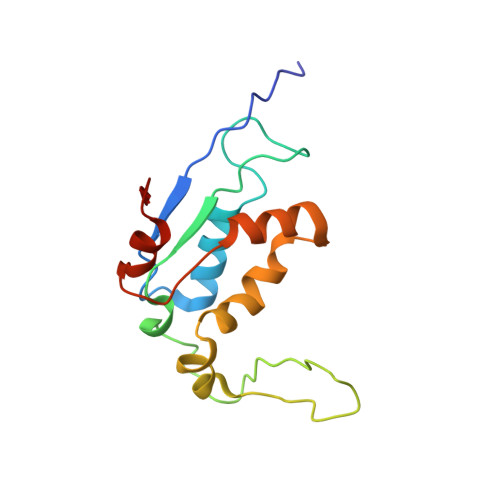
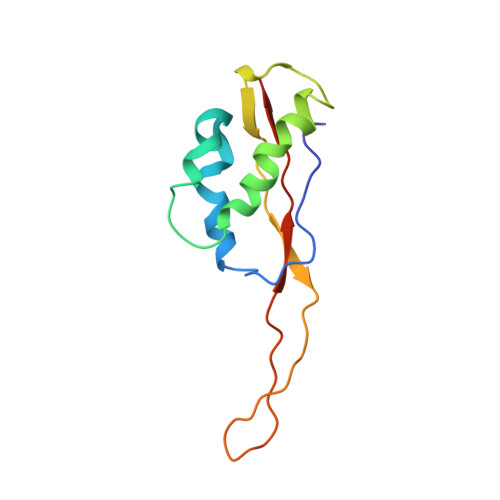
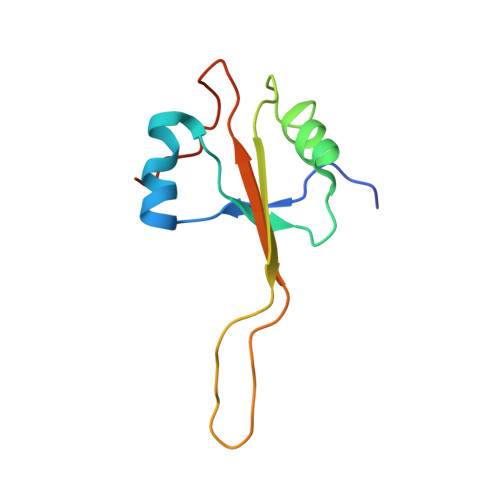
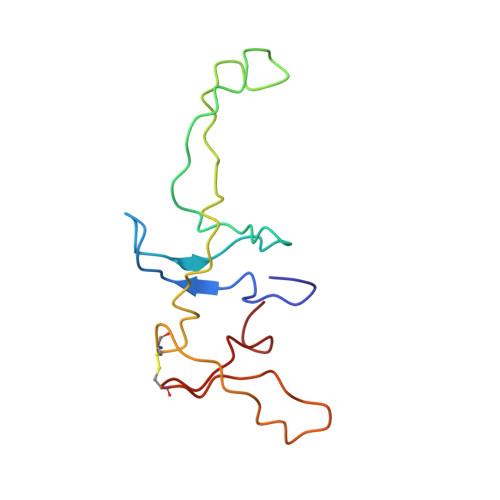
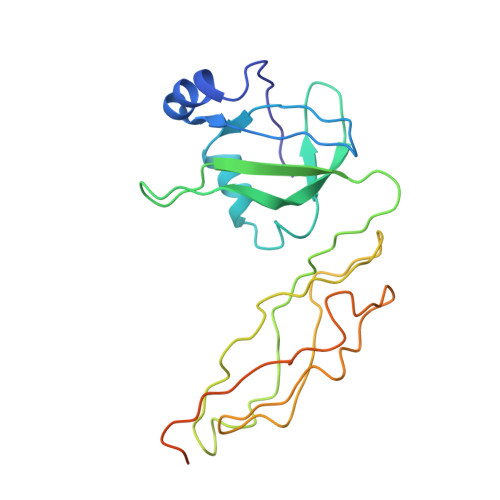
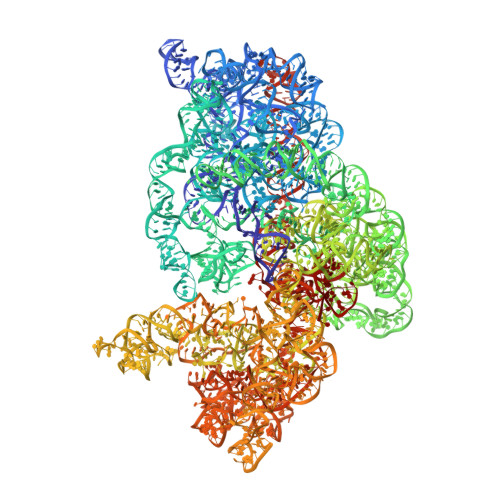
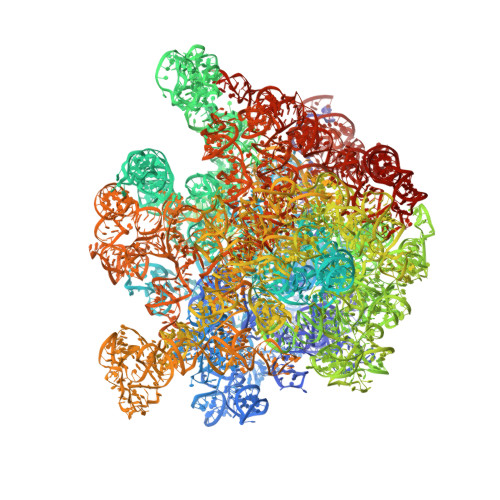
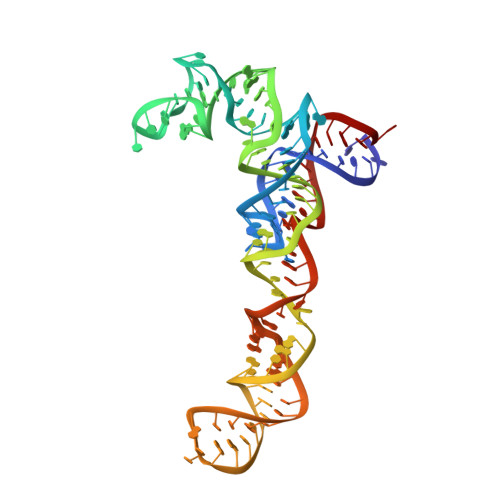
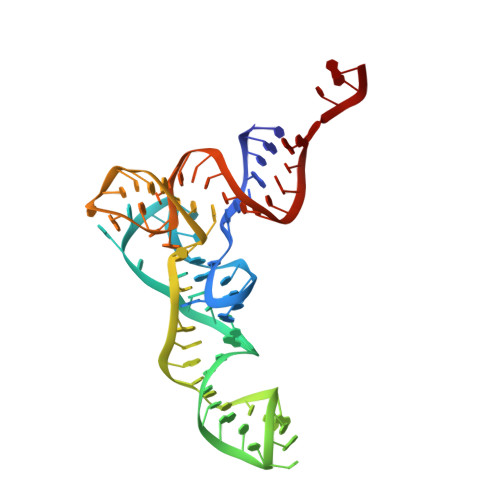
Structure of the GTP Form of Elongation Factor 4 (EF4) Bound to the Ribosome
Kumar, V., Ero, R., Ahmed, T., Goh, K.J., Zhan, Y., Bhushan, S., Gao, Y.G.(2016) J Biological Chem 291: 12943-12950
- PubMed: 27137929 Search on PubMedSearch on PubMed Central
- DOI: https://doi.org/10.1074/jbc.M116.725945
- Primary Citation Related Structures:
5IMQ, 5IMR - PubMed Abstract:
Elongation factor 4 (EF4) is a member of the family of ribosome-dependent translational GTPase factors, along with elongation factor G and BPI-inducible protein A. Although EF4 is highly conserved in bacterial, mitochondrial, and chloroplast genomes, its exact biological function remains controversial. Here we present the cryo-EM reconstitution of the GTP form of EF4 bound to the ribosome with P and E site tRNAs at 3.8-Å resolution. Interestingly, our structure reveals an unrotated ribosome rather than a clockwise-rotated ribosome, as observed in the presence of EF4-GDP and P site tRNA. In addition, we also observed a counterclockwise-rotated form of the above complex at 5.7-Å resolution. Taken together, our results shed light on the interactions formed between EF4, the ribosome, and the P site tRNA and illuminate the GTPase activation mechanism at previously unresolved detail.
- From the Institute of Molecular and Cell Biology, The Agency for Science, Technology and Research (A*STAR), 61 Biopolis Drive, 138673 Singapore, the School of Biological Sciences, Nanyang Technological University, 60 Nanyang Drive, 637551 Singapore, and.
Organizational Affiliation: