Transcription of lncRNA prt, clustered prt RNA sites for Mmi1 binding, and RNA polymerase II CTD phospho-sites govern the repression of pho1 gene expression under phosphate-replete conditions in fission yeast.
Chatterjee, D., Sanchez, A.M., Goldgur, Y., Shuman, S., Schwer, B.(2016) RNA 22: 1011-1025
- PubMed: 27165520 Search on PubMedSearch on PubMed Central
- DOI: https://doi.org/10.1261/rna.056515.116
- Primary Citation Related Structures:
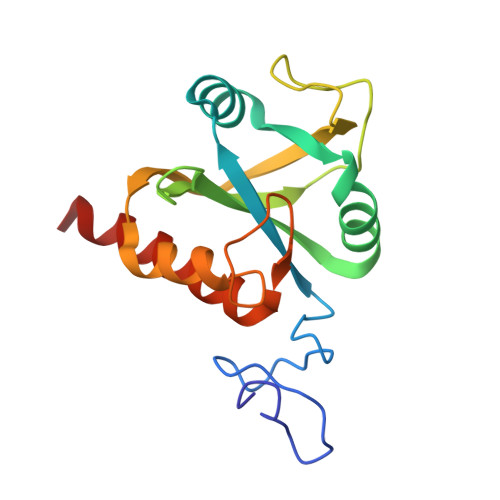
5H8A, 5HFZ - PubMed Abstract:
Expression of fission yeast Pho1 acid phosphatase is repressed during growth in phosphate-rich medium. Repression is mediated by transcription of the prt locus upstream of pho1 to produce a long noncoding (lnc) prt RNA. Repression is also governed by RNA polymerase II CTD phosphorylation status, whereby inability to place a Ser7-PO4 mark (as in S7A) derepresses Pho1 expression, and inability to place a Thr4-PO4 mark (as in T4A) hyper-represses Pho1 in phosphate replete cells. Here we find that basal pho1 expression from the prt-pho1 locus is inversely correlated with the activity of the prt promoter, which resides in a 110-nucleotide DNA segment preceding the prt transcription start site. CTD mutations S7A and T4A had no effect on the activity of the prt promoter or the pho1 promoter, suggesting that S7A and T4A affect post-initiation events in prt lncRNA synthesis that make it less and more repressive of pho1, respectively. prt lncRNA contains clusters of DSR (determinant of selective removal) sequences recognized by the YTH-domain-containing protein Mmi1. Altering the nucleobase sequence of two DSR clusters in the prt lncRNA caused hyper-repression of pho1 in phosphate replete cells, concomitant with increased levels of the prt transcript. The isolated Mmi1 YTH domain binds to RNAs with single or tandem DSR elements, to the latter in a noncooperative fashion. We report the 1.75 Å crystal structure of the Mmi1 YTH domain and provide evidence that Mmi1 recognizes DSR RNA via a binding mode distinct from that of structurally homologous YTH proteins that recognize m(6)A-modified RNA.
- Molecular Biology and Structural Biology Programs, Sloan-Kettering Institute, New York, New York 10065, USA.
Organizational Affiliation: