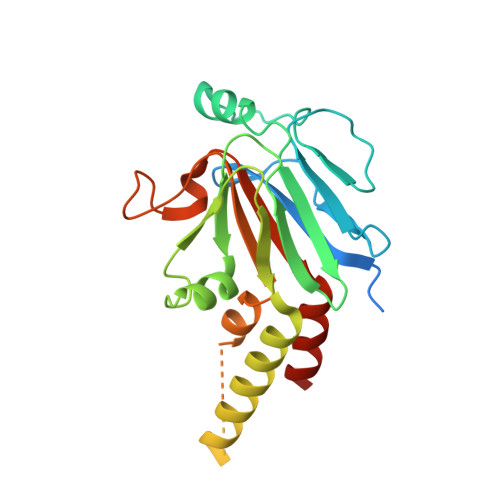
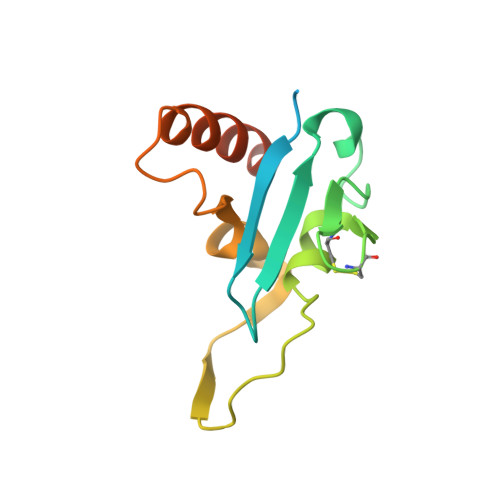
SnoN Stabilizes the SMAD3/SMAD4 Protein Complex.
Wallden, K., Nyman, T., Hallberg, B.M.(2017) Sci Rep 7: 46370-46370
- PubMed: 28397834 Search on PubMedSearch on PubMed Central
- DOI: https://doi.org/10.1038/srep46370
- Primary Citation Related Structures:
5C4V - PubMed Abstract:
TGF-β signaling regulates cellular processes such as proliferation, differentiation and apoptosis through activation of SMAD transcription factors that are in turn modulated by members of the Ski-SnoN family. In this process, Ski has been shown to negatively modulate TGF-β signaling by disrupting active R-SMAD/Co-SMAD heteromers. Here, we show that the related regulator SnoN forms a stable complex with the R-SMAD (SMAD3) and the Co-SMAD (SMAD4). To rationalize this stabilization at the molecular level, we determined the crystal structure of a complex between the SAND domain of SnoN and the MH2-domain of SMAD4. This structure shows a binding mode that is compatible with simultaneous coordination of R-SMADs. Our results show that SnoN, and SMAD heteromers can form a joint structural core for the binding of other transcription modulators. The results are of fundamental importance for our understanding of the molecular mechanisms behind the modulation of TGF-β signaling.
- Department of Cell and Molecular Biology, Karolinska Institutet, 171 77 Stockholm, Sweden.
Organizational Affiliation: