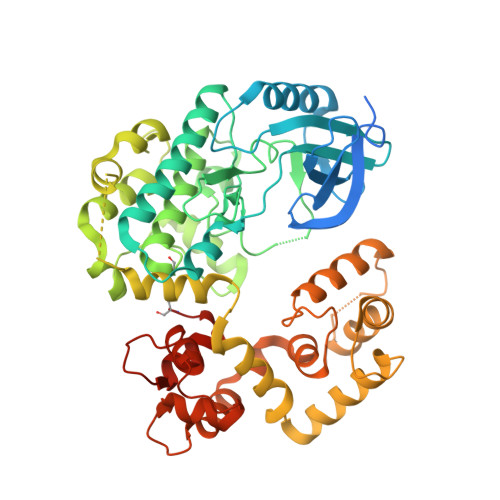
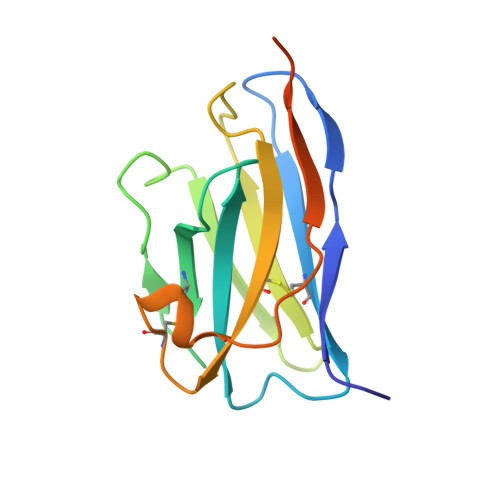
Allosteric activation of apicomplexan calcium-dependent protein kinases.
Ingram, J.R., Knockenhauer, K.E., Markus, B.M., Mandelbaum, J., Ramek, A., Shan, Y., Shaw, D.E., Schwartz, T.U., Ploegh, H.L., Lourido, S.(2015) Proc Natl Acad Sci U S A 112: E4975-E4984
- PubMed: 26305940 Search on PubMedSearch on PubMed Central
- DOI: https://doi.org/10.1073/pnas.1505914112
- Primary Citation Related Structures:
4YGA - PubMed Abstract:
Calcium-dependent protein kinases (CDPKs) comprise the major group of Ca2+-regulated kinases in plants and protists. It has long been assumed that CDPKs are activated, like other Ca2+-regulated kinases, by derepression of the kinase domain (KD). However, we found that removal of the autoinhibitory domain from Toxoplasma gondii CDPK1 is not sufficient for kinase activation. From a library of heavy chain-only antibody fragments (VHHs), we isolated an antibody (1B7) that binds TgCDPK1 in a conformation-dependent manner and potently inhibits it. We uncovered the molecular basis for this inhibition by solving the crystal structure of the complex and simulating, through molecular dynamics, the effects of 1B7-kinase interactions. In contrast to other Ca2+-regulated kinases, the regulatory domain of TgCDPK1 plays a dual role, inhibiting or activating the kinase in response to changes in Ca2+ concentrations. We propose that the regulatory domain of TgCDPK1 acts as a molecular splint to stabilize the otherwise inactive KD. This dependence on allosteric stabilization reveals a novel susceptibility in this important class of parasite enzymes.
- Whitehead Institute for Biomedical Research, Cambridge, MA 02142;
Organizational Affiliation: