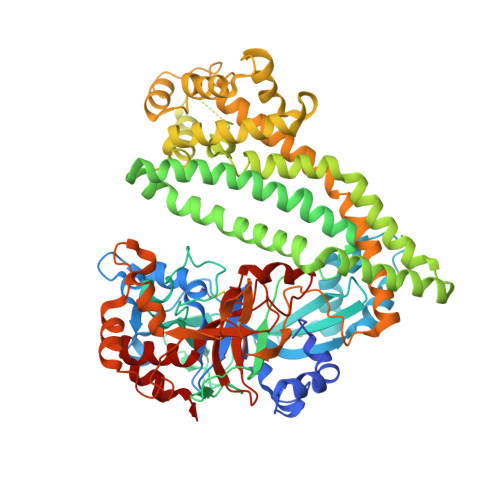
Structural and mutational analyses of dipeptidyl peptidase 11 from Porphyromonas gingivalis reveal the molecular basis for strict substrate specificity.
Sakamoto, Y., Suzuki, Y., Iizuka, I., Tateoka, C., Roppongi, S., Fujimoto, M., Inaka, K., Tanaka, H., Yamada, M., Ohta, K., Gouda, H., Nonaka, T., Ogasawara, W., Tanaka, N.(2015) Sci Rep 5: 11151-11151
- PubMed: 26057589 Search on PubMedSearch on PubMed Central
- DOI: https://doi.org/10.1038/srep11151
- Primary Citation Related Structures:
4XZY, 4Y01, 4Y02, 4Y04, 4Y06 - PubMed Abstract:
The dipeptidyl peptidase 11 from Porphyromonas gingivalis (PgDPP11) belongs to the S46 family of serine peptidases and preferentially cleaves substrates with Asp/Glu at the P1 position. The molecular mechanism underlying the substrate specificity of PgDPP11, however, is unknown. Here, we report the crystal structure of PgDPP11. The enzyme contains a catalytic domain with a typical double β-barrel fold and a recently identified regulatory α-helical domain. Crystal structure analyses, docking studies, and biochemical studies revealed that the side chain of Arg673 in the S1 subsite is essential for recognition of the Asp/Glu side chain at the P1 position of the bound substrate. Because S46 peptidases are not found in mammals and the Arg673 is conserved among DPP11s, we anticipate that DPP11s could be utilised as targets for antibiotics. In addition, the present structure analyses could be useful templates for the design of specific inhibitors of DPP11s from pathogenic organisms.
- School of Pharmacy, Iwate Medical University, 2-1-1 Nishitokuta, Yahaba, Iwate 028-3694, Japan.
Organizational Affiliation: