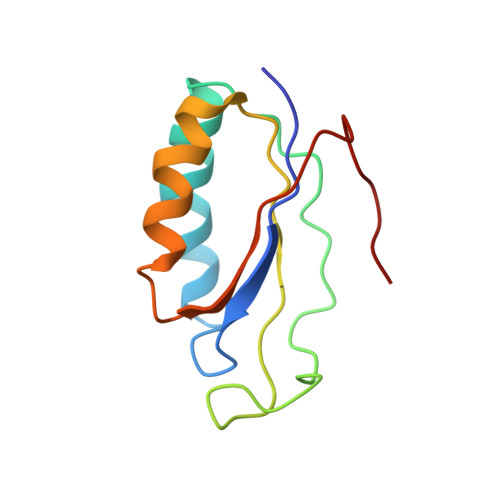
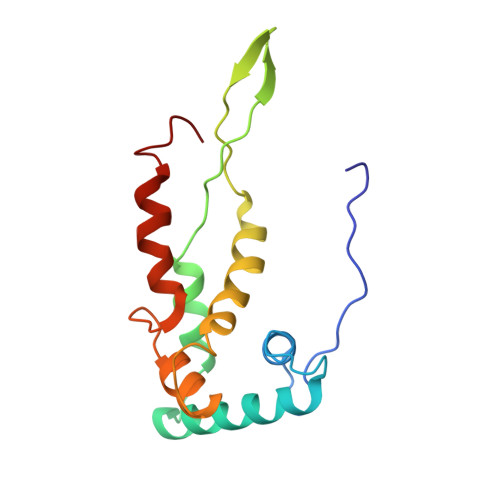
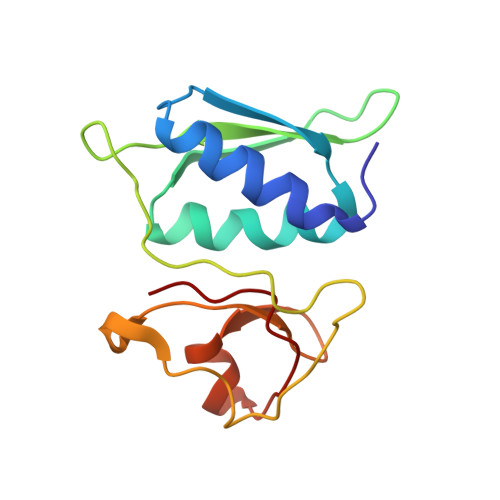
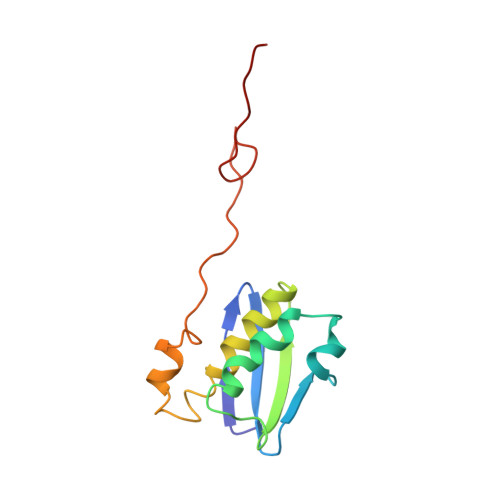
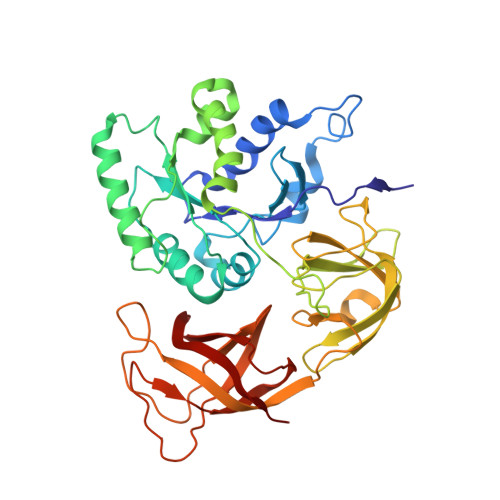
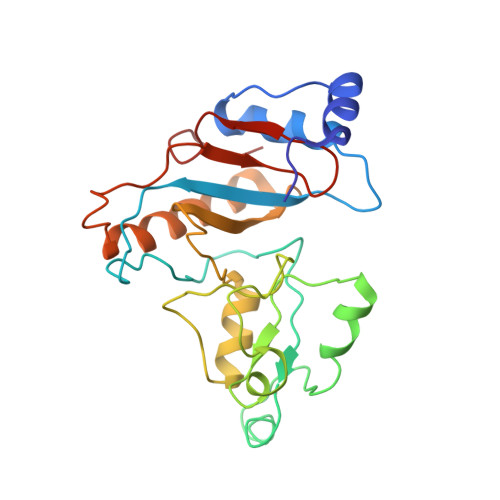
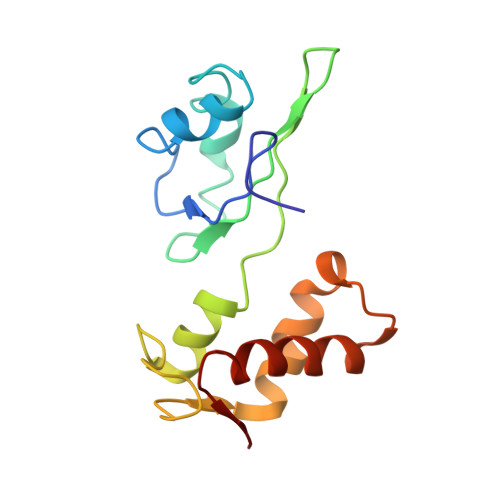
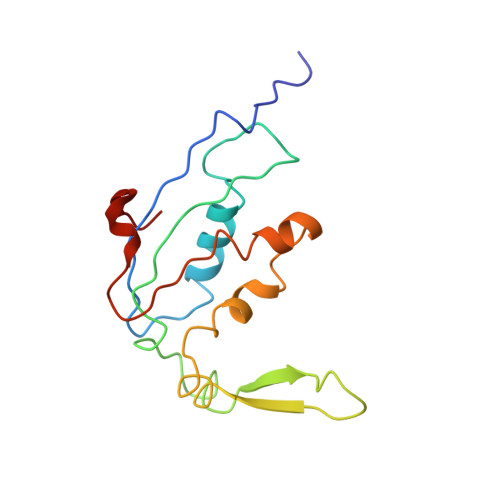
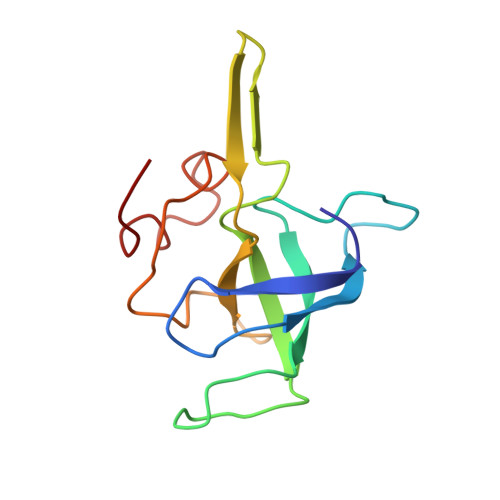
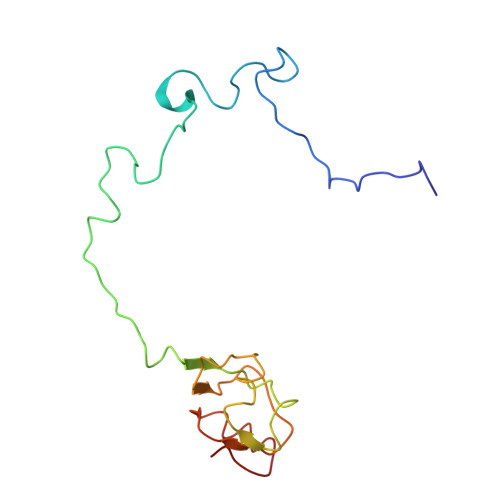
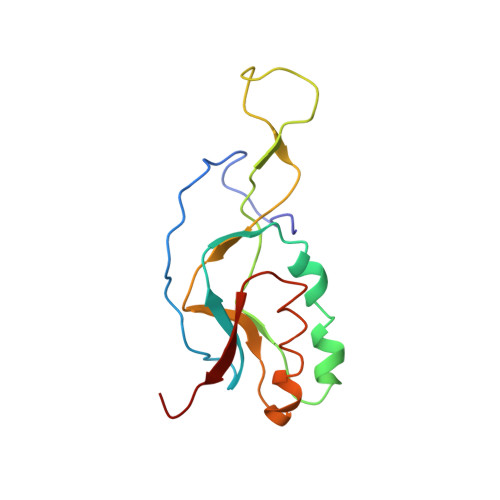
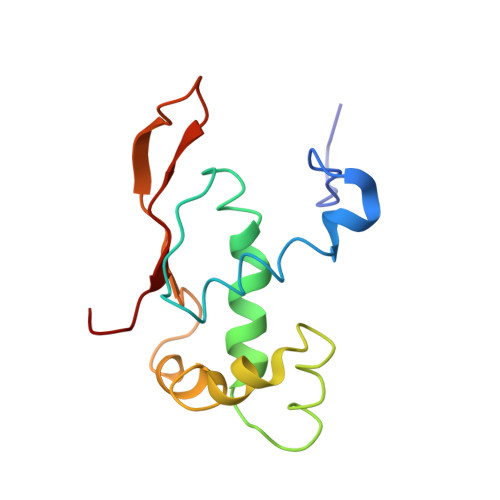
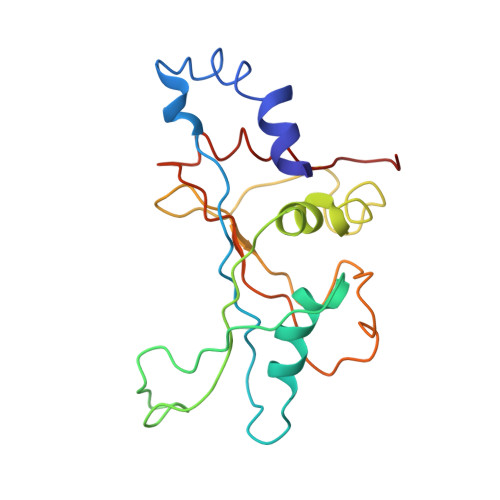
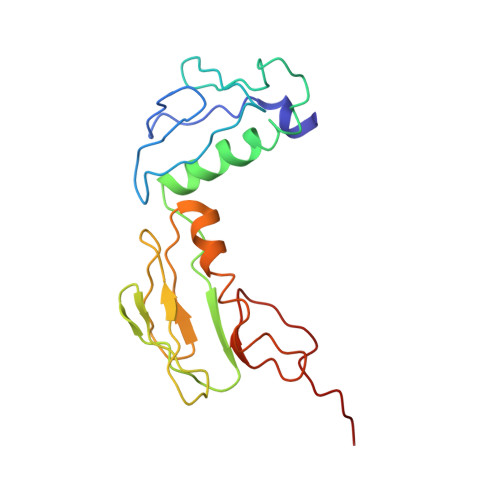
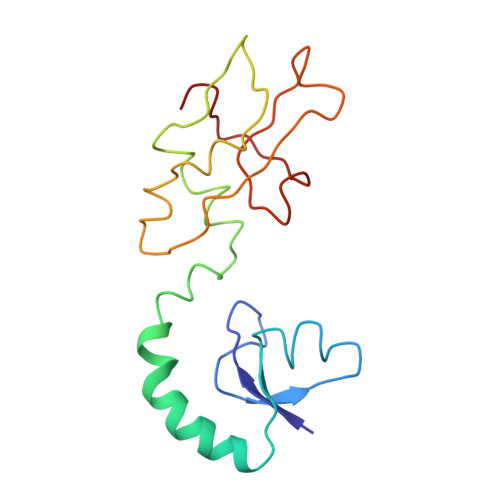
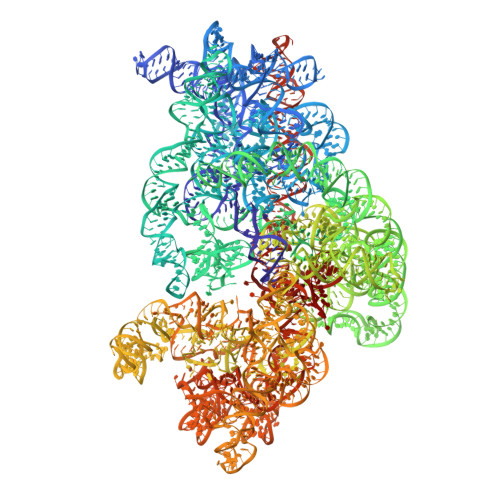
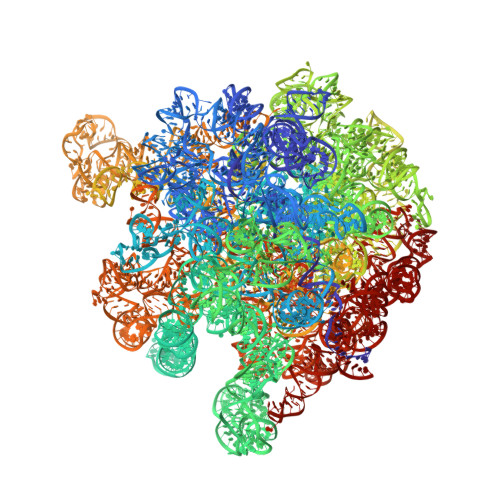
Ribosome-induced changes in elongation factor Tu conformation control GTP hydrolysis
Villa, E., Sengupta, J., Trabuco, L.G., LeBarron, J., Baxter, W.T., Shaikh, T.R., Grassucci, R.A., Nissen, P., Ehrenberg, M., Schulten, K., Frank, J.(2009) Proc Natl Acad Sci U S A 106: 1063-1068
- PubMed: 19122150 Search on PubMedSearch on PubMed Central
- DOI: https://doi.org/10.1073/pnas.0811370106
- Primary Citation Related Structures:
4V69 - PubMed Abstract:
In translation, elongation factor Tu (EF-Tu) molecules deliver aminoacyl-tRNAs to the mRNA-programmed ribosome. The GTPase activity of EF-Tu is triggered by ribosome-induced conformational changes of the factor that play a pivotal role in the selection of the cognate aminoacyl-tRNAs. We present a 6.7-A cryo-electron microscopy map of the aminoacyl-tRNA x EF-Tu x GDP x kirromycin-bound Escherichia coli ribosome, together with an atomic model of the complex obtained through molecular dynamics flexible fitting. The model reveals the conformational changes in the conserved GTPase switch regions of EF-Tu that trigger hydrolysis of GTP, along with key interactions, including those between the sarcin-ricin loop and the P loop of EF-Tu, and between the effector loop of EF-Tu and a conserved region of the 16S rRNA. Our data suggest that GTP hydrolysis on EF-Tu is controlled through a hydrophobic gate mechanism.
- Beckman Institute, University of Illinois at Urbana-Champaign, Urbana, IL 61801, USA.
Organizational Affiliation: