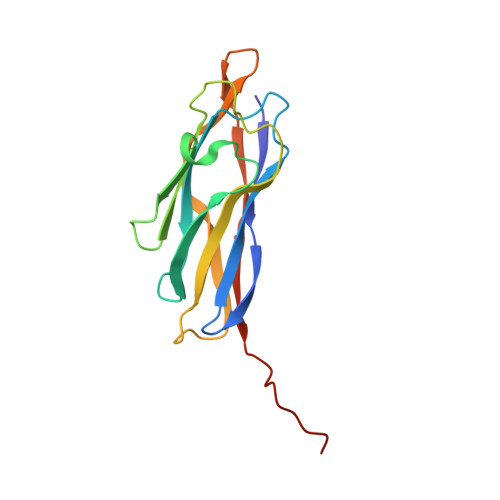
Combined Crystal Structure of a Type-I Cohesin, Mutation and Affinity-Binding Studies Reveal Structural Determinants of Cohesin-Dockerin Specificity
Cameron, K., Alves, V.D., Bayer, E.A., Gilbert, H.J., Ferreira, L.M.A., Fontes, C.M.G.A., Najmudin, S.(2015) J Biological Chem 290: 16215
- PubMed: 25934389 Search on PubMedSearch on PubMed Central
- DOI: https://doi.org/10.1074/jbc.M115.653303
- Primary Citation Related Structures:
4UMS - PubMed Abstract:
Cohesin-dockerin interactions orchestrate the assembly of one of nature's most elaborate multienzyme complexes, the cellulosome. Cellulosomes are produced exclusively by anaerobic microbes and mediate highly efficient hydrolysis of plant structural polysaccharides, such as cellulose and hemicellulose. In the canonical model of cellulosome assembly, type I dockerin modules of the enzymes bind to reiterated type I cohesin modules of a primary scaffoldin. Each type I dockerin contains two highly conserved cohesin-binding sites, which confer quaternary flexibility to the multienzyme complex. The scaffoldin also bears a type II dockerin that anchors the entire complex to the cell surface by binding type II cohesins of anchoring scaffoldins. In Bacteroides cellulosolvens, however, the organization of the cohesin-dockerin types is reversed, whereby type II cohesin-dockerin pairs integrate the enzymes into the primary scaffoldin, and type I modules mediate cellulosome attachment to an anchoring scaffoldin. Here, we report the crystal structure of a type I cohesin from B. cellulosolvens anchoring scaffoldin ScaB to 1.84-Å resolution. The structure resembles other type I cohesins, and the putative dockerin-binding site, centered at β-strands 3, 5, and 6, is likely to be conserved in other B. cellulosolvens type I cohesins. Combined computational modeling, mutagenesis, and affinity-based binding studies revealed similar hydrogen-bonding networks between putative Ser/Asp recognition residues in the dockerin at positions 11/12 and 45/46, suggesting that a dual-binding mode is not exclusive to the integration of enzymes into primary cellulosomes but can also characterize polycellulosome assembly and cell-surface attachment. This general approach may provide valuable structural information of the cohesin-dockerin interface, in lieu of a definitive crystal structure.
- From the CIISA, Faculdade de Medicina Veterinária, Universidade de Lisboa, Avenida da Universidade Técnica, 1300-477 Lisboa, Portugal.
Organizational Affiliation: