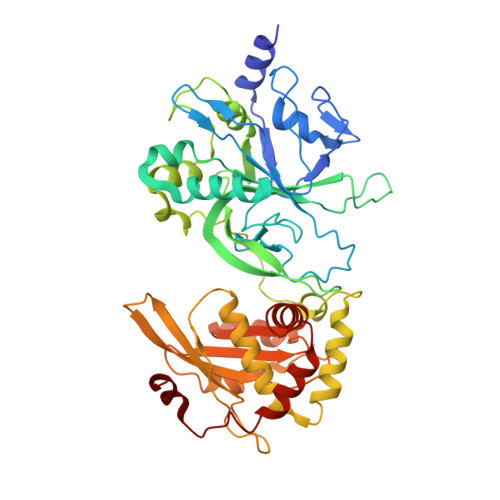
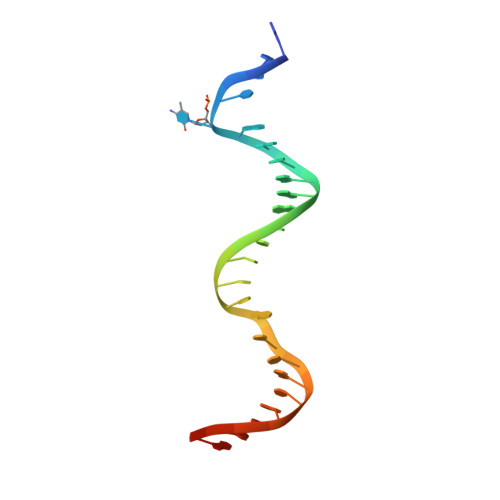
Modification-dependent restriction endonuclease, MspJI, flips 5-methylcytosine out of the DNA helix.
Horton, J.R., Wang, H., Mabuchi, M.Y., Zhang, X., Roberts, R.J., Zheng, Y., Wilson, G.G., Cheng, X.(2014) Nucleic Acids Res 42: 12092-12101
- PubMed: 25262349 Search on PubMedSearch on PubMed Central
- DOI: https://doi.org/10.1093/nar/gku871
- Primary Citation Related Structures:
4R28 - PubMed Abstract:
MspJI belongs to a family of restriction enzymes that cleave DNA containing 5-methylcytosine (5mC) or 5-hydroxymethylcytosine (5hmC). MspJI is specific for the sequence 5(h)mC-N-N-G or A and cleaves with some variability 9/13 nucleotides downstream. Earlier, we reported the crystal structure of MspJI without DNA and proposed how it might recognize this sequence and catalyze cleavage. Here we report its co-crystal structure with a 27-base pair oligonucleotide containing 5mC. This structure confirms that MspJI acts as a homotetramer and that the modified cytosine is flipped from the DNA helix into an SRA-like-binding pocket. We expected the structure to reveal two DNA molecules bound specifically to the tetramer and engaged with the enzyme's two DNA-cleavage sites. A coincidence of crystal packing precluded this organization, however. We found that each DNA molecule interacted with two adjacent tetramers, binding one specifically and the other non-specifically. The latter interaction, which prevented cleavage-site engagement, also involved base flipping and might represent the sequence-interrogation phase that precedes specific recognition. MspJI is unusual in that DNA molecules are recognized and cleaved by different subunits. Such interchange of function might explain how other complex multimeric restriction enzymes act.
- Department of Biochemistry, Emory University School of Medicine, 1510 Clifton Road, Atlanta, GA 30322, USA.
Organizational Affiliation: