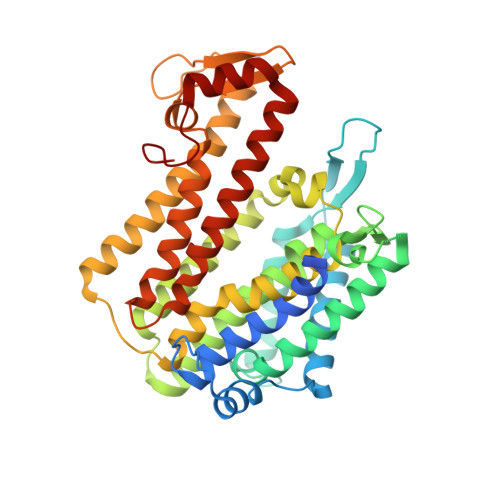
Structure of an integral membrane sterol reductase from Methylomicrobium alcaliphilum.
Li, X., Roberti, R., Blobel, G.(2015) Nature 517: 104-107
- PubMed: 25307054 Search on PubMedSearch on PubMed Central
- DOI: https://doi.org/10.1038/nature13797
- Primary Citation Related Structures:
4QUV - PubMed Abstract:
Sterols are essential biological molecules in the majority of life forms. Sterol reductases including Δ(14)-sterol reductase (C14SR, also known as TM7SF2), 7-dehydrocholesterol reductase (DHCR7) and 24-dehydrocholesterol reductase (DHCR24) reduce specific carbon-carbon double bonds of the sterol moiety using a reducing cofactor during sterol biosynthesis. Lamin B receptor (LBR), an integral inner nuclear membrane protein, also contains a functional C14SR domain. Here we report the crystal structure of a Δ(14)-sterol reductase (MaSR1) from the methanotrophic bacterium Methylomicrobium alcaliphilum 20Z (a homologue of human C14SR, LBR and DHCR7) with the cofactor NADPH. The enzyme contains ten transmembrane segments (TM1-10). Its catalytic domain comprises the carboxy-terminal half (containing TM6-10) and envelops two interconnected pockets, one of which faces the cytoplasm and houses NADPH, while the other one is accessible from the lipid bilayer. Comparison with a soluble steroid 5β-reductase structure suggests that the reducing end of NADPH meets the sterol substrate at the juncture of the two pockets. A sterol reductase activity assay proves that MaSR1 can reduce the double bond of a cholesterol biosynthetic intermediate, demonstrating functional conservation to human C14SR. Therefore, our structure as a prototype of integral membrane sterol reductases provides molecular insight into mutations in DHCR7 and LBR for inborn human diseases.
- Laboratory of Cell Biology, Howard Hughes Medical Institute, The Rockefeller University, New York, New York 10065, USA.
Organizational Affiliation: