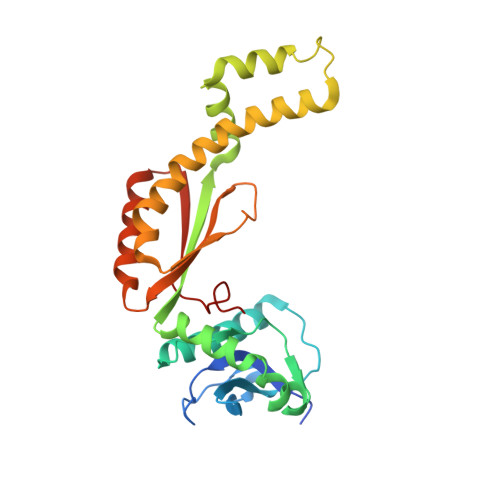
Structure of the gas vesicle protein GvpF from the cyanobacterium Microcystis aeruginosa.
Xu, B.Y., Dai, Y.N., Zhou, K., Liu, Y.T., Sun, Q., Ren, Y.M., Chen, Y., Zhou, C.Z.(2014) Acta Crystallogr D Biol Crystallogr 70: 3013-3022
- PubMed: 25372690 Search on PubMed
- DOI: https://doi.org/10.1107/S1399004714021312
- Primary Citation Related Structures:
4QSG - PubMed Abstract:
Gas vesicles are gas-filled proteinaceous organelles that provide buoyancy for bacteria and archaea. A gene cluster that is highly conserved in various species encodes about 8-14 proteins (Gvp proteins) that are involved in the formation of gas vesicles. Here, the first crystal structure of the gas vesicle protein GvpF from Microcystis aeruginosa PCC 7806 is reported at 2.7 Å resolution. GvpF is composed of two structurally distinct domains (the N-domain and C-domain), both of which display an α+β class overall structure. The N-domain adopts a novel fold, whereas the C-domain has a modified ferredoxin fold with an apparent variation owing to an extension region consisting of three sequential helices. The two domains pack against each other via interactions with a C-terminal tail that is conserved among cyanobacteria. Taken together, it is concluded that the overall architecture of GvpF presents a novel fold. Moreover, it is shown that GvpF is most likely to be a structural protein that is localized at the gas-facing surface of the gas vesicle by immunoblotting and immunogold labelling-based tomography.
- Hefei National Laboratory for Physical Sciences at the Microscale and School of Life Sciences, University of Science and Technology of China, Heifei, Anhui 230027, People's Republic of China.
Organizational Affiliation: