Identification of Polyketide Inhibitors Targeting 3-Dehydroquinate Dehydratase in the Shikimate Pathway of Enterococcus faecalis
Cheung, V.W., Xue, B., Hernandez-Valladares, M., Go, M.K., Tung, A., Aguda, A.H., Robinson, R.C., Yew, W.S.(2014) PLoS One 9: e103598-e103598
- PubMed: 25072253
- DOI: https://doi.org/10.1371/journal.pone.0103598
- Primary Citation Related Structures:
4PH6 - PubMed Abstract:
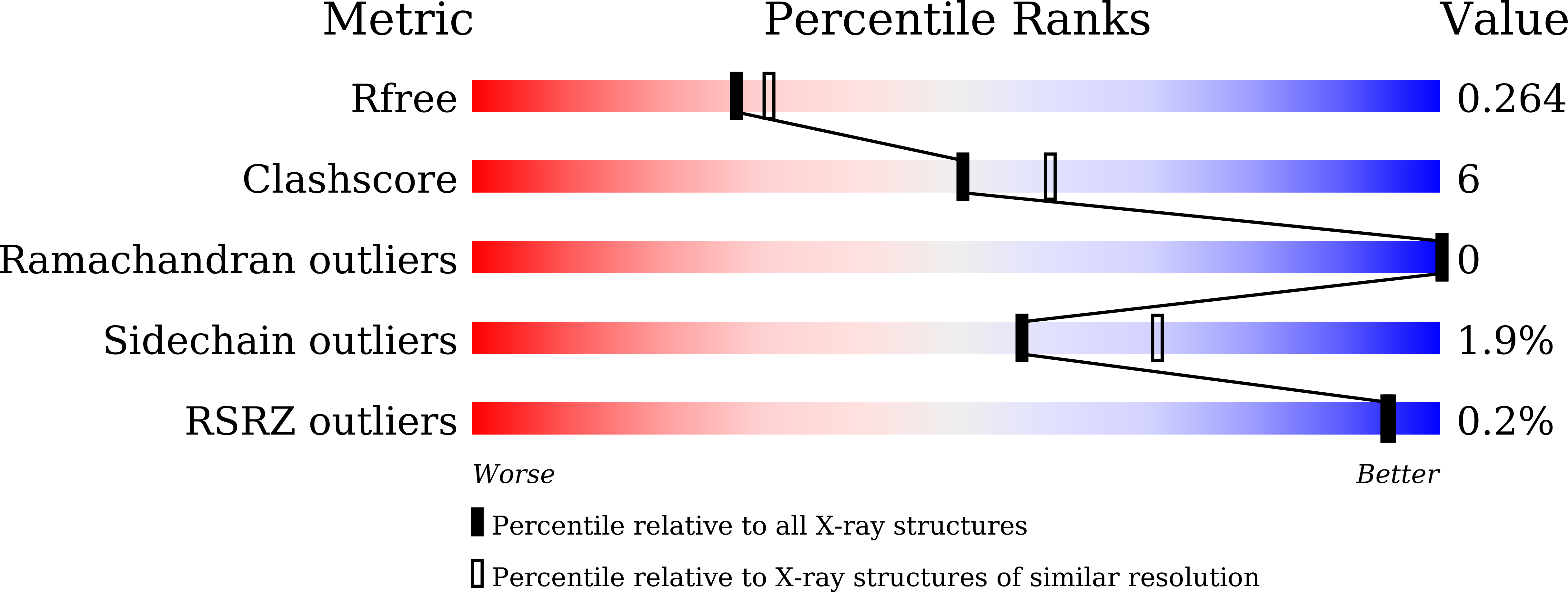
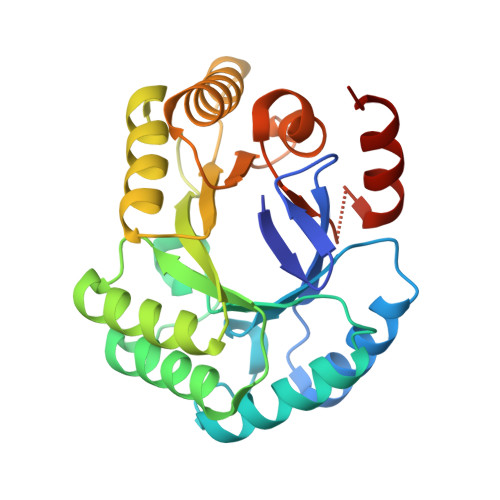
Due to the emergence of resistance toward current antibiotics, there is a pressing need to develop the next generation of antibiotics as therapeutics against infectious and opportunistic diseases of microbial origins. The shikimate pathway is exclusive to microbes, plants and fungi, and hence is an attractive and logical target for development of antimicrobial therapeutics. The Gram-positive commensal microbe, Enterococcus faecalis, is a major human pathogen associated with nosocomial infections and resistance to vancomycin, the "drug of last resort". Here, we report the identification of several polyketide-based inhibitors against the E. faecalis shikimate pathway enzyme, 3-dehydroquinate dehydratase (DHQase). In particular, marein, a flavonoid polyketide, both inhibited DHQase and retarded the growth of Enterococcus faecalis. The purification, crystallization and structural resolution of recombinant DHQase from E. faecalis (at 2.2 Å resolution) are also reported. This study provides a route in the development of polyketide-based antimicrobial inhibitors targeting the shikimate pathway of the human pathogen E. faecalis.
- Department of Biochemistry, Yong Loo Lin School of Medicine, National University of Singapore, Singapore, Singapore.
Organizational Affiliation: